Lime production experience 4.0
Italian plant engineering contractor QualiCal has designed and built a 150 t/d Synthesis regenerative vertical kiln (PFR – twin shaft burning unit) for Köhler Kalk. The kiln boasts rock-bottom investment and running costs and is capable of turning out quicklime of the highest quality. Digitization was one of the project’s major elements.
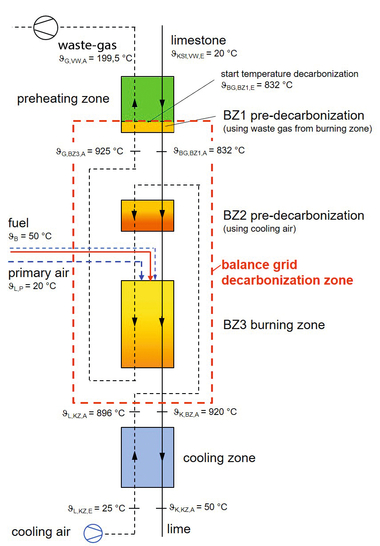
1 The calcination process
The calcination of limestone is a chemical process of decomposition in which heat applied to carbonates decomposes them into oxides. Decomposition takes place at a temperature of around 810 °C:
CaCO3 + heat = CaO + CO2
To ensure that the limestone rock decomposes all the way through, the heat of dissociation must be able to penetrate to the core. So, for complete calcination, the surface of the stone must be heated to above 900 °C. The surface temperature, however, should not exceed 1100 – 1150 °C, because that would cause sintering, or dead burning, of the calcined...