Controlled hydration of burnt limes and cementitious by-products using a novel laboratory planetary mixer
Lime hydration is a fundamental process in the production of calcium and magnesium hydroxides, widely used in construction materials, environmental applications, and industrial processes such as flue gas desulfurization and wastewater treatment [1–3]. The hydration behaviour of burnt limes is controlled by both intrinsic material properties—such as chemical composition, mineralogy, porosity, and reactivity—and process parameters including water dosage, temperature evolution, pressure conditions, and mixing dynamics. These factors directly influence phase conversion, as well as the development of key technological properties such as BET specific surface area, pore structure, and product stability.
Burnt limes are produced by calcination of carbonate raw materials using different kiln technologies, including rotary kilns and parallel flow regenerative (PFR) kilns, also known as twin-shaft regenerative (TSR) kilns. Among these technologies, PFR kilns are currently considered the most efficient systems for producing highly reactive quicklime with low specific energy consumption and reduced environmental impact [4–10]. Depending on the raw material and burning conditions, quicklime and dolomitic lime may exhibit significant variability in chemical composition, mineralogy, porosity and slaking reactivity, which in turn affects hydration behaviour and final product performance [1–3].
In industrial practice, hydration is carried out in multi-stage hydrators designed to produce a wide range of products, from standard hydrated limes for construction applications to high-performance sorbents for environmental uses [1, 2, 11, 12]. The process complexity is further increased by the growing use of heterogeneous materials such as dolomitic limes, natural hydraulic limes, and supplementary cementitious materials (SCM) by-products, including fly ash and bottom ash, which are increasingly exploited within circular economy strategies [3, 13–17]. These materials introduce additional variability in hydration kinetics and phase evolution, making process control and optimization more challenging.
Despite the widespread industrial use of lime hydrators, laboratory-scale systems capable of reproducing the key thermodynamic and kinetic conditions of industrial hydrators remain limited. Conventional laboratory systems, such as Dewar flasks and stirred reactors, do not allow simultaneous control of temperature, pressure, water dosage and mixing dynamics. As a result, they fail to accurately simulate industrial operating conditions, limiting their applicability for process optimization, raw material screening, and prediction of product performance.
To overcome these limitations, a purpose-built fully integrated planetary mixer was developed at Cimprogetti’s technological laboratories to simulate industrial lime hydration conditions under controlled laboratory-scale conditions. The system enables precise control of temperature, pressure, water dosage, and mixing parameters, allowing reproducible and process-oriented hydration testing across a wide range of materials.
The present study aims to evaluate the performance of this system through controlled hydration experiments on quicklime, dolomitic lime, natural hydraulic lime, and SCM by-products. Specific hydration protocols were developed to achieve targeted product properties for both construction and environmental applications. The resulting materials were characterized in terms of phase composition by X-ray diffraction (XRD) with Rietveld refinement [18–20] and porosimetric properties (BET surface area, pore volume, and pore size distribution) [21, 22], with the objective of establishing a direct link between process conditions, hydration mechanisms, and final product performance.
To the authors’ knowledge, this study represents a novel approach to reproduce industrial lime hydration conditions at laboratory scale by simultaneously controlling pressure, temperature, water dosage and mixing dynamics using a purpose-built planetary mixer.
2 The purpose-built fully integrated planetary mixer
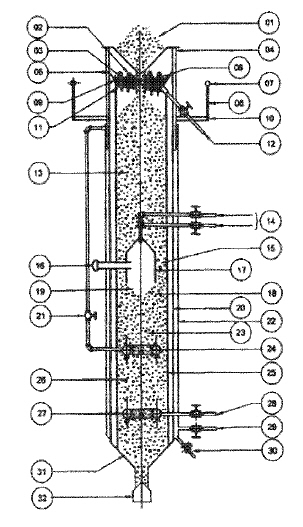
To reproduce industrial hydration conditions under controlled laboratory-scale environments, a purpose-built fully integrated planetary mixer was developed. This novel mixer (Figure 1) consists of several sets of integrated mechanical and thermal control components. Its main components include an electric control panel, a vacuum pump, mixing shafts with blades, and 3 kW heating elements for temperature control. Temperature control is ensured by a platinum probe (PT100) strategically positioned at the tank’s base. Meanwhile, the heating elements can be finely tuned to maintain temperature. Pressure management is controlled via an inlet connected to a compressor, allowing for adjustments up to a maximum of 2500 mbar-g. The system maintains constant pressure during operation. Compressed air is used to regulate pressure and remove excess vapors after water addition. The introduction of demineralized water, facilitated through the hopper, can occur either during system operation under standard ambient conditions or subsequent to depressurization by the vacuum pump. Vacuum operation enables controlled water transfer from the hopper to the tank and supports steam retention during dolomitic lime hydration. This process is fundamental in sustaining the crucial reaction steam necessary for the hydration of dolomitic limestone. At its core, the mixer comprises a cylindrical tank with a capacity of 5 L and a diameter of 200 mm (hydration chamber in Figure 1b). Mixing is accomplished through the dynamic interaction of two blades performing a controlled motion along the tank’s perimeter while rotating around their axis. This mixing configuration ensures uniform material distribution within the tank volume. Additionally, the incorporation of a side scraping paddle enhances efficiency by retrieving any material adhering to the walls, ensuring an efficient and uniform mixing process. The combination of controlled temperature, pressure and mixing conditions makes the system suitable for simulating key operating parameters of industrial lime hydrators at laboratory scale.

Figure 1 Experimental setup of the laboratory planetary mixer: (a) general view of the mixer and its main components, including water dosing system, control panel, and 5 L tank; (b) internal view of the tank showing the mixer chamber, air inlet, and pressure/vacuum connections; (c) mixing system, including shafts, blades, and side scraping paddle; (d) control panel interface
3.1 Materials supply and source
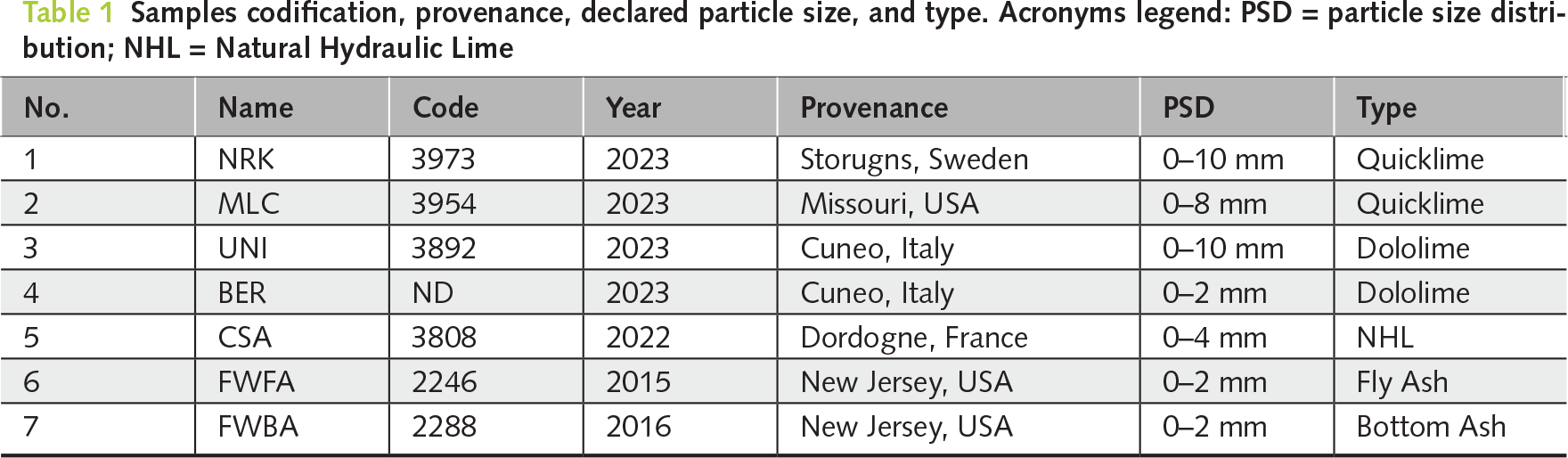
The experimental design was developed to reproduce representative industrial hydration conditions under controlled laboratory conditions. Five burnt limes and two SCM by-products were supplied by different international producers in the typical particle size fractions used in industrial hydration plants. The investigated materials comprise: 1) a quicklime from Storugns, Sweden (NRK); 2) a high-calcium lime, slowly reactive, from Missouri, USA (MLC); 3) and 4) two dolomitic limes from Cuneo, Italy (UNI and BER); 5) a natural hydraulic lime from Dordogne, France (CSA); 6) a fly ash from a power plant in New Jersey, USA (FWFA), and, finally, 7) a bottom ash from New Jersey, USA (FWBA). Samples codes, provenance, and particle size distribution (PSD) are reported in Table 1.
3.2 Physicochemical tests and slaking reactivity
The available lime index (ALI) was used as a key parameter to assess hydration potential and process behaviour and was determined using the sugar method [23]. Slaking reactivity was determined according to the European standard for building limes [24]. Moisture content after hydration was determined by a moisture analyzer, while real or absolute density was determined using a gas pycnometer.
3.3 Hydration tests in the planetary mixer
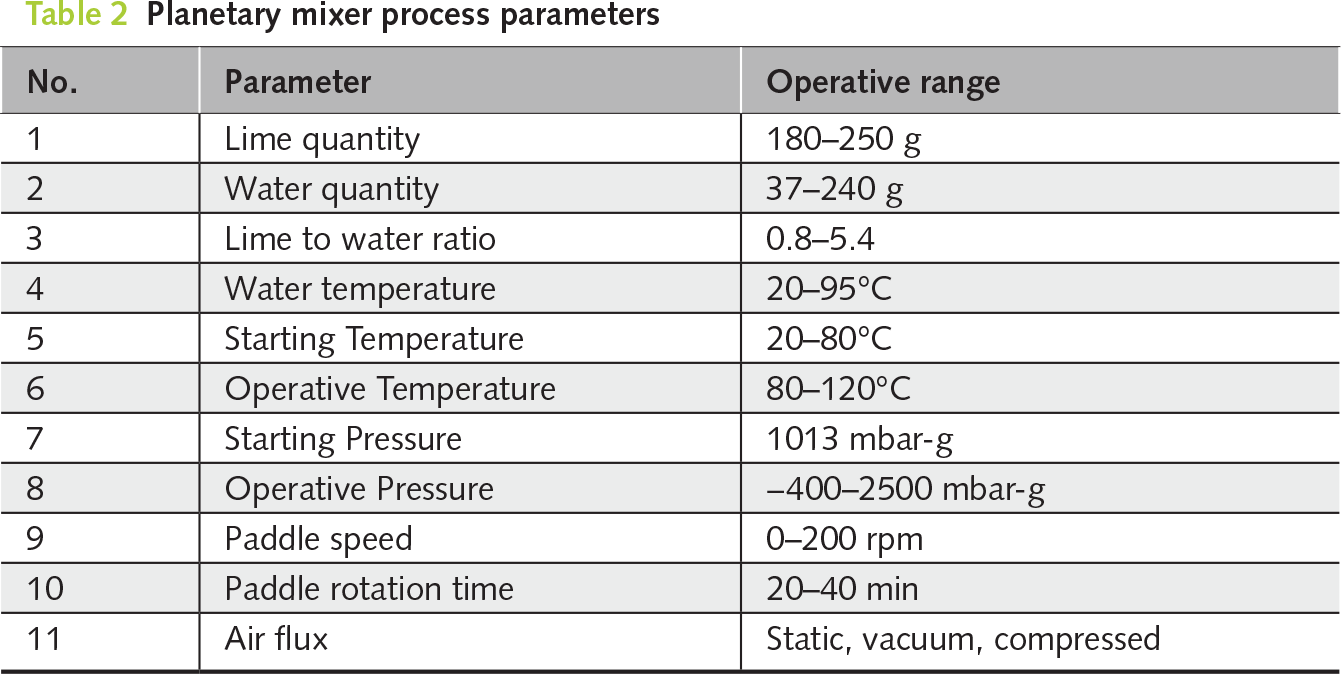
Dry hydration tests were performed using the new fully integrated planetary mixer (Figure 1). The process parameters were adjusted according to the type of raw material to optimize product performance. The main variables included water dosage, initial and operating temperature, pressure conditions (static, fluxing, or compressed air), and mixing parameters (paddle speed and rotation time). These parameters directly control hydration behaviour in terms of phase development and BET surface area. The selected operating ranges and testing approach (Table 2) were designed to reproduce typical industrial hydrator operating conditions, enabling a direct process-oriented interpretation of the results.
3.4 X-ray powder diffraction and nitrogen gas adsorption analyses
The X-ray diffraction (XRD) analysis was performed for phase identification and quantification using the Rietveld method [18, 19] through Profex software [20]. Nitrogen adsorption was used to determine BET surface area and pore structure, including pore-volume (PV) and pore-size (PS) distributions [21, 22].
4.1 High calcium limes or quicklimes
Two high-calcium quicklimes (NRK and MLC) were hydrated using the new planetary mixer. The sample NRK was found to be moderately reactive (i.e., ΔT 40 or t60 = 3.3 min; Tmax = 69.8°C). Conversely, the sample MLC was slowly reactive (i.e., ΔT 40 or t60 = 36.6 min, Tmax = 62.5°C), although ALI values are similar in both samples (≈85 wt%) (refer to Table 3 and Figure 2a,b). QPA results indicate that both samples are dominated by lime (CaO: 85.7–92.3 wt%), with only minor secondary phases and traces of portlandite related to slight pre-hydration (see Table 4, and Figure 3a–c).
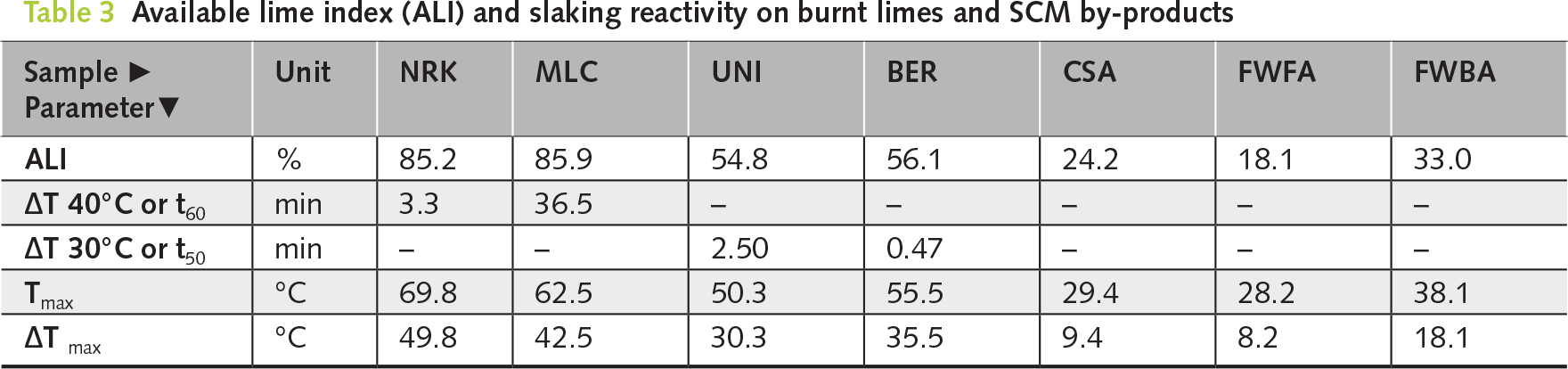
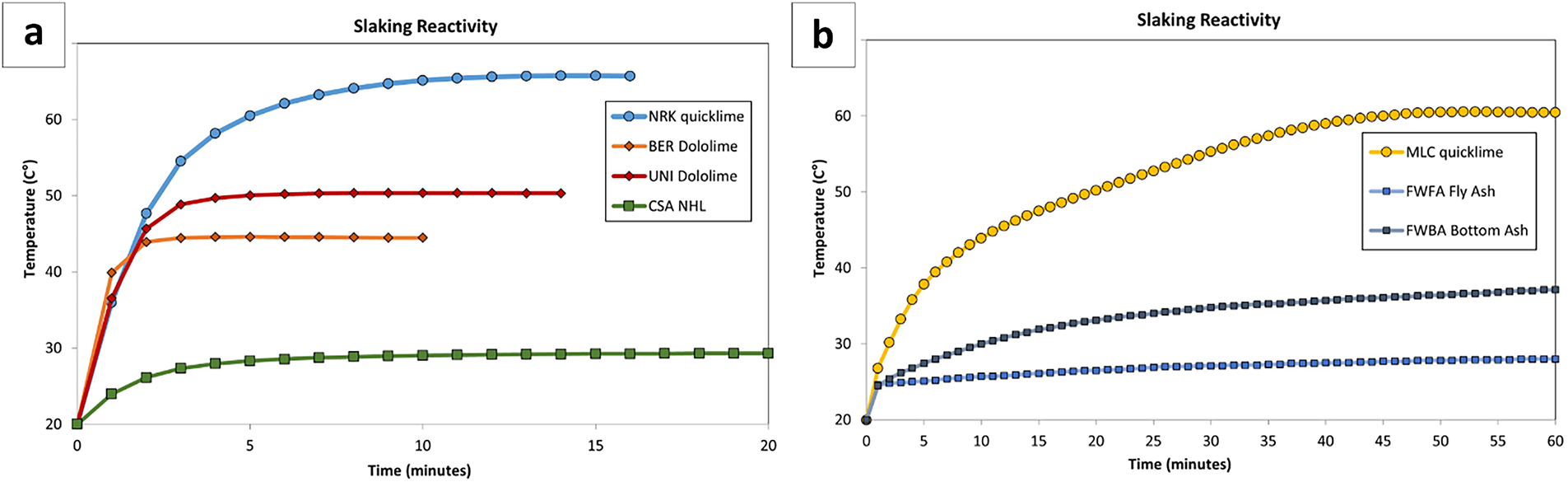
Figure 2 Slaking reactivity plots. The variation in testing duration reflects the different hydration kinetics of the investigated samples, whereas the final reaction temperature is primarily governed by their free CaO content
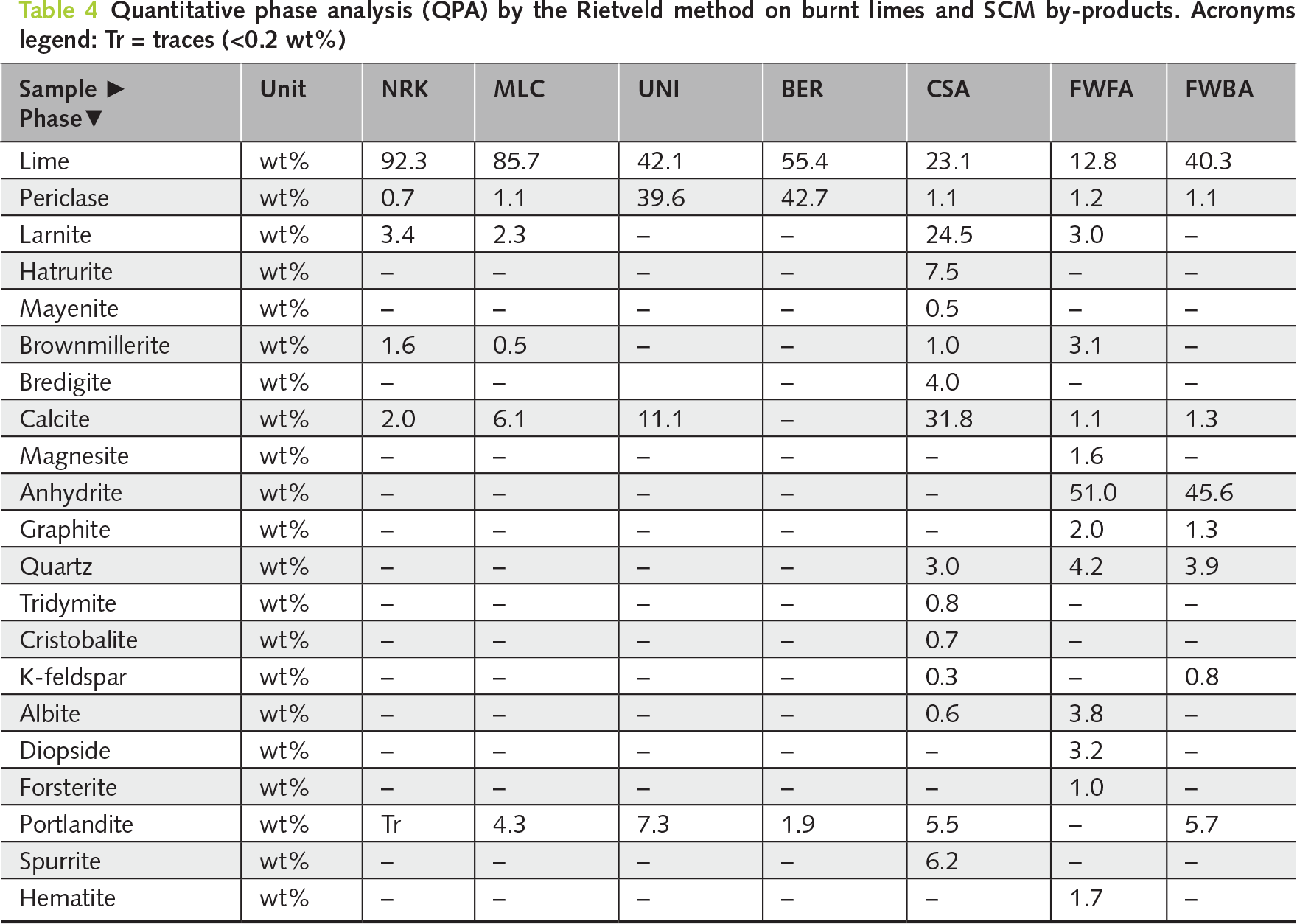
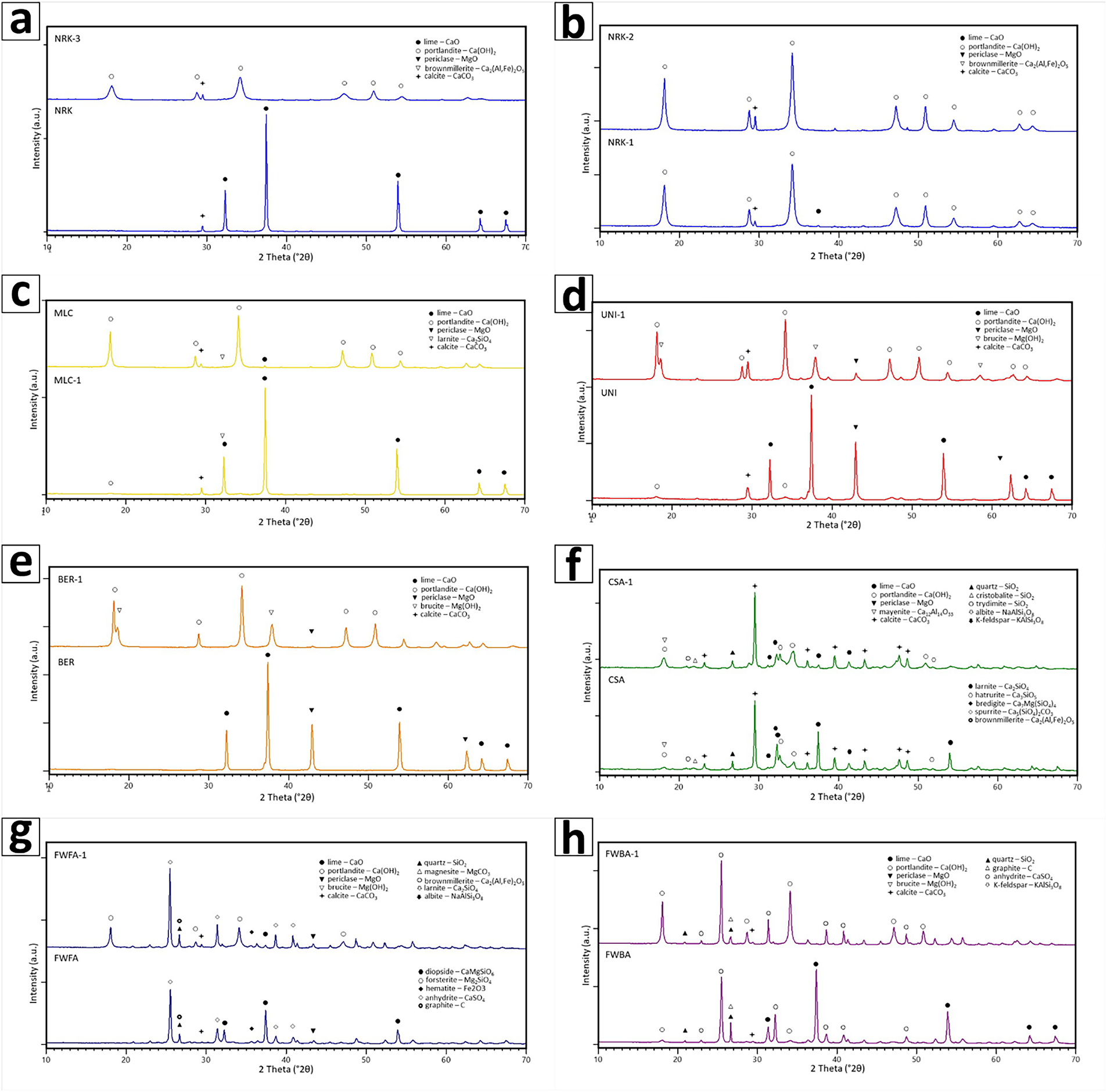
Figure 3 XRD patterns of burnt and hydrated samples: (a–c) quicklimes (NRK, MLC), (d, e) dolomitic limes (UNI, BER), and (f–h) NHL and SCM by-products (CSA, FWFA, FWBA), showing phase evolution from oxides (CaO, MgO) to hydroxides (portlandite, brucite) after hydration
Two standard hydration recipes were defined for construction-grade hydrated lime: one for moderately reactive quicklime (NRK-1; W/L = 0.83) and one for slowly reactive quicklime (MLC-1; W/L = 1.0). The higher water dosage adopted for MLC-1 was necessary to achieve full hydration of the less reactive lime.
Two additional recipes were developed to produce high-BET hydrated lime for FGD applications: one based on water excess (NRK-2; W/L = 1.33), resulting in a wet product requiring drying, and one based on stoichiometric hydration with TEA additive (NRK-3; W/L = 0.69; TEA = 0.8 wt%), aimed at enhancing surface area under drier conditions (Table 5).
The following process parameters were kept constant across all recipes for quicklime: 20 min of reaction time, 25°C of water temperature, ambient operative pressure, and 200 rpm as paddle speed.
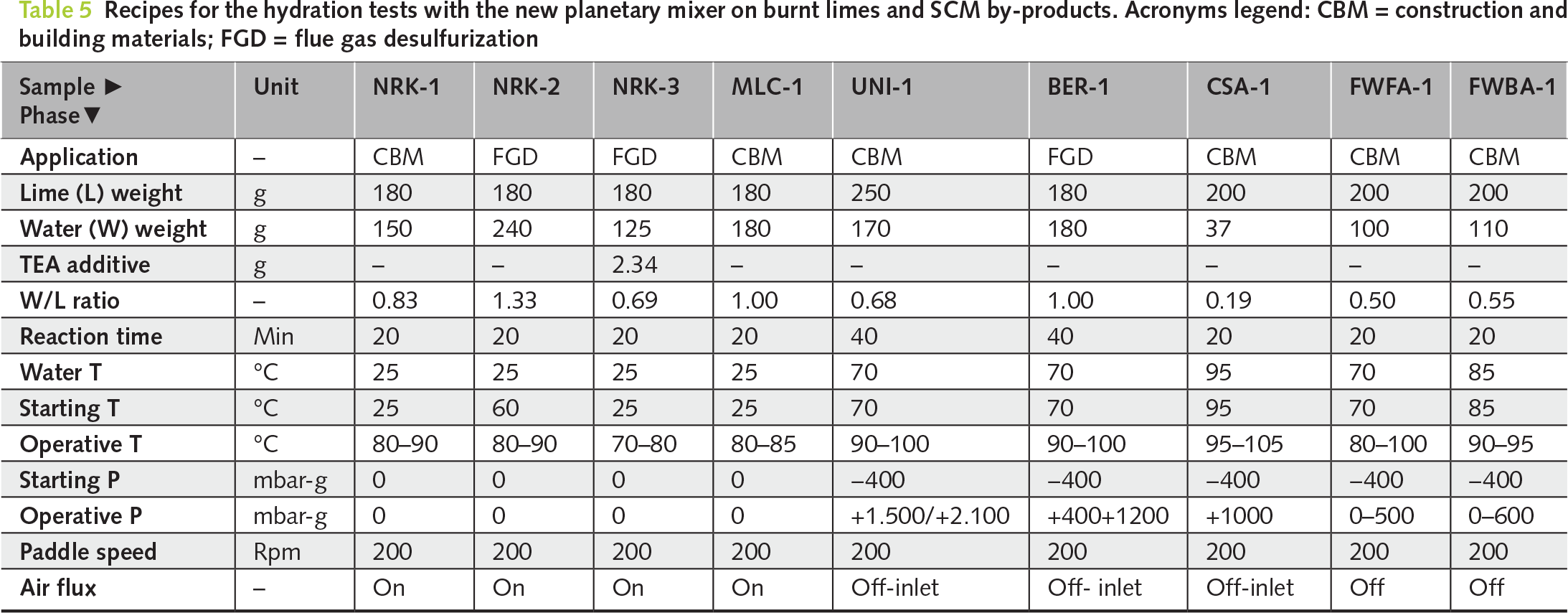
Subsequently, hydrated lime samples (NRK-1, NRK-2, NRK-3, and MLC-1) were analysed by XRD. The quantitative phase analysis (QPA) revealed the extent of oxide-hydroxide conversion, thereby providing an indicator of product quality (Table 6; Figure 3b–c).
QPA of the hydrated products confirmed near-complete conversion of CaO to portlandite, which became the dominant phase in all samples (90.5–96.5 wt%). Minor calcite is mainly related to recarbonation, while residual lime and other secondary phases remained negligible. Porosimetric analyses showed clear differences among the hydration recipes (Table 7; Figures 4 and 5). BET surface area ranged from 20.8 m2/g (MLC-1) to 35.4 m2/g (NRK-2). Pore volume ranged from 0.075 cm3/g (NRK-3) to 0.173 cm3/g (NRK-2), while pore diameter varied between 7.5 and 16.2 nm. Real density remained within a narrow range (2.21–2.29 g/cm3). These results indicate that hydration conditions influence both phase conversion and porosimetric properties.
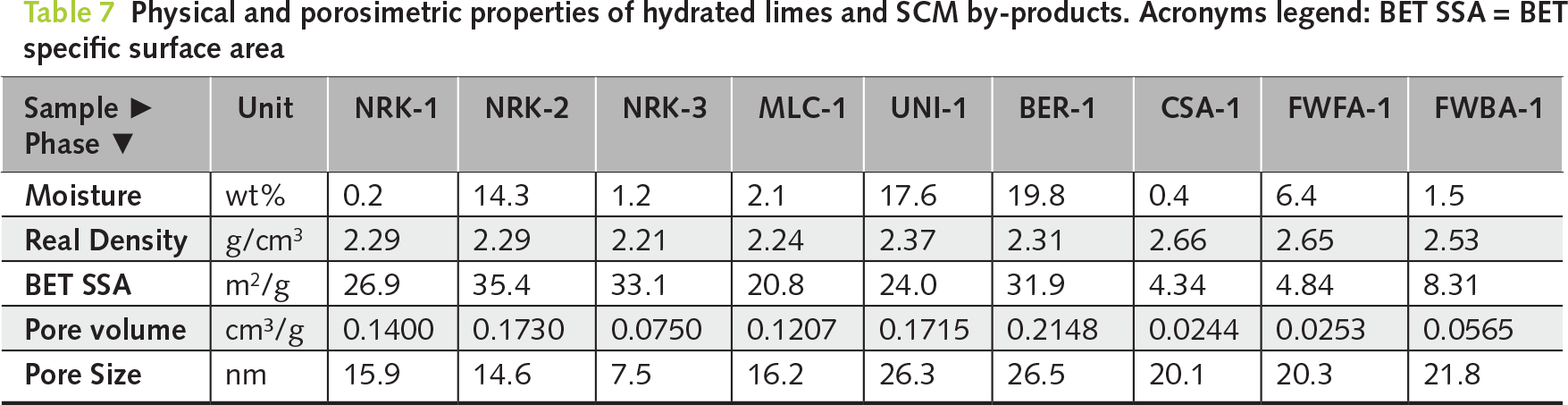
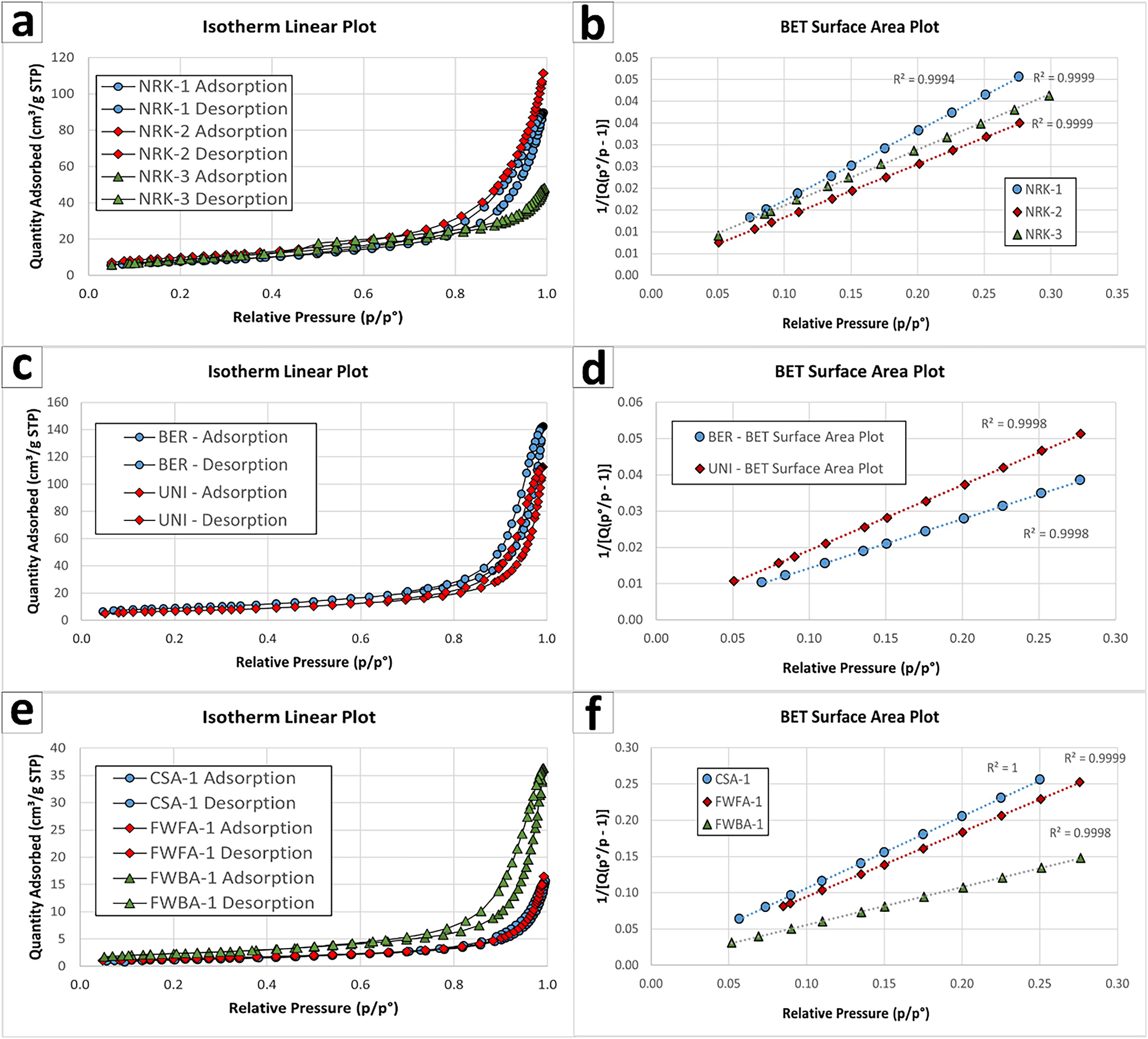
Figure 4 Nitrogen adsorption–desorption isotherms and BET plots of selected hydrated samples (NRK-1, NRK-2, NRK-3, MLC-1, UNI-1, BER-1, CSA-1, FWFA-1 and FWBA-1), illustrating differences in surface area development as a function of hydration route and raw material type
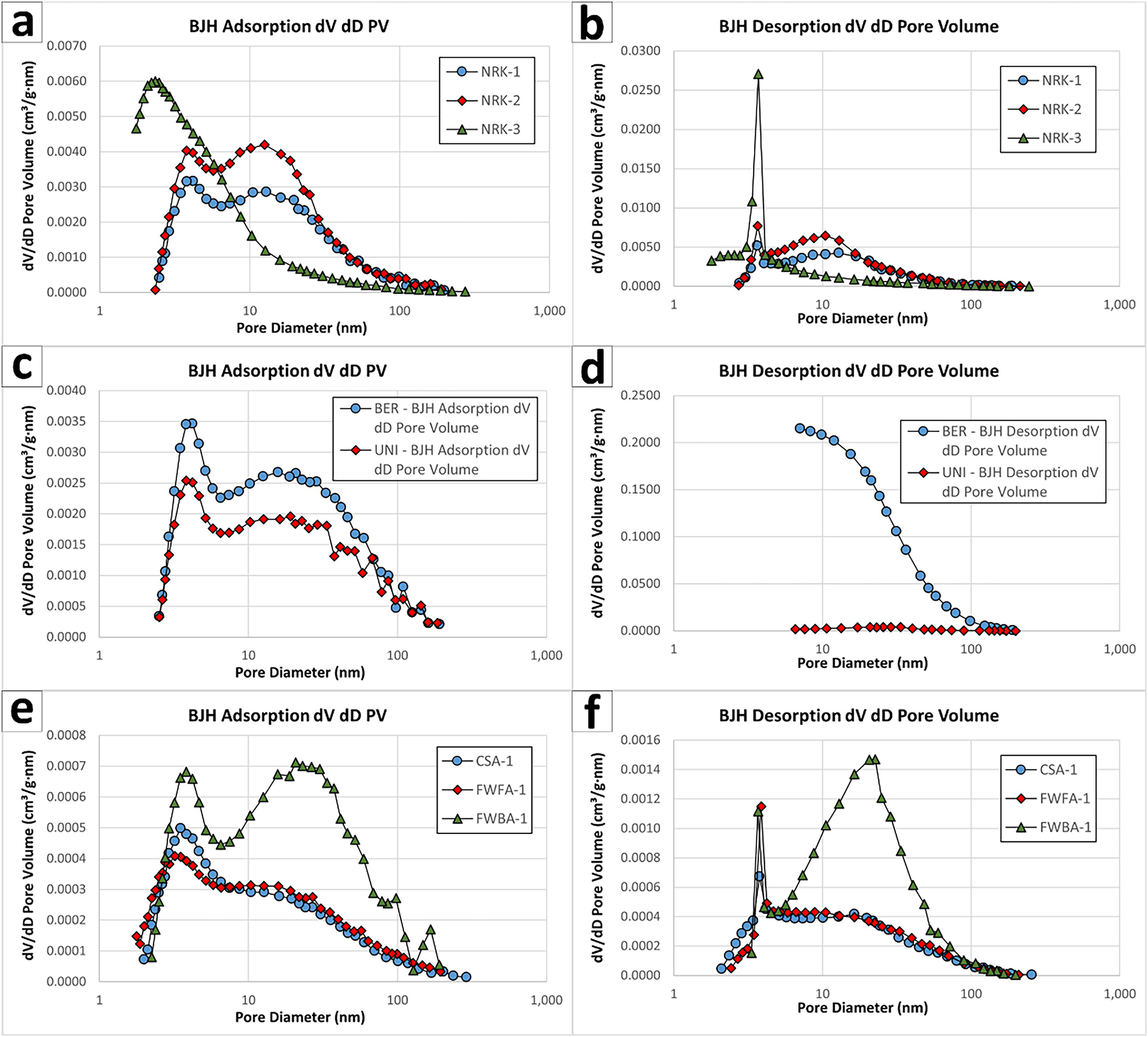
Figure 5 BJH pore size distribution curves of hydrated samples, showing the effect of hydration route and material type on pore structure and mesoporosity development
4.2 Magnesium-rich lime or dololime
Two dolomitic limes (UNI and BER) with different reactivity were hydrated using the planetary mixer. UNI showed moderate reactivity (t50 = 2.5 min; Tmax = 50.3°C), whereas BER was highly reactive (t50 = 0.5 min; Tmax = 55.5°C), with comparable available lime index values (ALI ≈ 55 wt%; Table 3; Figure 2a,b).
A pressure-controlled hydration protocol was applied to promote the formation of double-hydrated dolomitic lime. The selected conditions included a water-to-lime ratio between 0.68 and 1.0, elevated temperature (90–100°C), and controlled pressure evolution up to ~2100 mbar-g (Table 5).
Hydration resulted in significant conversion of both CaO and MgO into portlandite (43.0–51.3 wt%) and brucite (40.5–47.5 wt%), confirming the effectiveness of pressure-assisted conditions in promoting MgO hydration (Table 6; Figure 3d,e). Porosimetric properties showed significant differences between the two samples. BER-1 exhibited higher BET surface area (~32 m2/g) and pore volume (~0.21 cm3/g), whereas UNI-1 showed lower values (~24 m2/g and ~0.17 cm3/g, respectively; Table 7, and Figures 4 and 5).
These results highlight that the combined influence of intrinsic material properties (e.g., reactivity and grain size) and process conditions plays a key role in controlling hydration efficiency and sorbent performance of dolomitic limes.
The natural hydraulic lime sample (CSA) showed very low reactivity (i.e., ΔTmax 9.3°C; ALI value is 24.2 wt%; Table 3 and Figure 2a), reflecting its limited content of free CaO. A specific hydration protocol was therefore adopted, using a low water-to-lime ratio (W/L = 0.19) and elevated temperature conditions (95°C), combined with controlled pressure (see Table 5).
Hydration resulted in only partial conversion to portlandite (~20 wt%), while the product retained a complex mineralogical composition dominated by calcite and silicate phases (Table 6 and Figure 3f). Porosimetric analysis confirmed low BET surface area (~4 m2/g) and relatively high density (2.66 g/cm³), consistent with the hydraulic nature of the material (Table 7 and Figures 4 and 5).
These results indicate that NHL behaves as a low-reactivity system under the tested conditions and may require more aggressive process conditions to enhance hydration efficiency.
4.4 Supplementary cementitious materials (SCM by-products)
The present study focuses on the hydration of CaO-bearing phases in SCM by-products rather than on their cementitious reactivity. This approach evaluates their behaviour as lime-based reactive systems rather than conventional pozzolanic materials. Both fly (FWFA) and bottom ash (FWBA) exhibited very low hydration reactivity (ΔTmax = 8.2–18.1°C; ALI = 18.1–33.0 wt%; Table 3 and Figure 2b), reflecting their limited content of free CaO.
A specific hydration protocol was applied using high water-to-lime ratios (W/L = 5.0–5.5), elevated temperatures (70–85°C), and controlled pressure conditions (Table 5).
Hydration led to partial conversion of CaO-bearing phases into portlandite (26.7–46.7 wt%), while the overall mineralogical composition remained dominated by anhydrite and other low-reactivity phases (Table 6; Figure 3g,h). Porosimetric properties were limited, with low BET surface area values (4.8–8.3 m2/g) and relatively high densities (2.53–2.65 g/cm³), indicating restricted development of reactive surface (Table 7; Figures 4 and 5).
These results indicate that SCM by-products behave as partially reactive systems under the tested conditions and cannot be treated as fully hydrable materials, in contrast to conventional limes.
5.1 Applications for green construction materials
Dry hydrated limes and hydrated SCM by-products intended for construction applications were produced using standard hydration protocols in the planetary mixer. These included moderately reactive quicklime (NRK-1), slowly reactive quicklime (MLC-1), natural hydraulic lime (CSA-1), and hydrated fly and bottom ashes (FWFA-1 and FWBA-1). The selected water-to-lime ratios were adjusted according to the free lime availability of each raw material. Standard quicklimes required W/L values between 0.83 and 1.0, whereas lower-ALI materials such as NHL and SCM by-products required adapted hydration conditions. This confirms that hydration behaviour is primarily controlled by available lime index (ALI) and slaking kinetics. Highly reactive limes undergo rapid conversion to portlandite under standard conditions, whereas low-reactivity materials require more demanding process parameters.
XRD-QPA data support this interpretation by documenting the conversion of CaO into portlandite after hydration (Tables 4–6, Figures 3a–c and 6).
Figure 6 Relationship between available lime index (ALI) and lime content determined by XRD-QPA
For construction applications, complete or near-complete CaO conversion is essential to minimize the risk of delayed expansion in mortars, fillers, and pre-mixed cementitious products. The applied hydration protocols reduced residual CaO in quicklimes and promoted portlandite formation in NHL and SCM by-products, although to a lesser extent in the latter. This confirms that the planetary mixer can be used not only for conventional lime hydration, but also for screening partially reactive industrial by-products for construction-oriented reuse.
From a practical perspective, the preservation of raw material freshness prior hydration is critical. Pre-hydration and storage alteration reduce both conversion efficiency and porosimetric development. Burnt lime and reactive by-products should therefore be stored under dry conditions and hydrated as close as possible to production. This aspect becomes particularly important when high-surface-area products are targeted.
Slowly reactive aerial limes such as MLC require specific operational adjustments, including higher water dosage and, potentially, longer residence time. From an industrial perspective, a short maturation stage in silo remains advisable to complete hydration and stabilize the final product, especially when residual moisture is kept within the typical 2–5 wt% range. This approach supports the valorization of low-reactive industrial by-products within a circular economy framework. Controlled hydration testing therefore represents a practical tool for raw material selection and process optimization in construction applications.
5.2 Applications for acid abatement and flue gas desulfurization
5.2.1 High-BET hydration of quicklime: process routes
High-BET hydrated limes for acid abatement and flue gas desulfurization were produced through two main quicklime-based routes: excess-water hydration (NRK-2) and additive-assisted hydration (NRK-3). A third route, based on pressure-controlled hydration of dolomitic lime (UNI-1 and BER-1) is discussed separately in Section 5.2.3.
The conventional route for producing high-BET hydrated limes is based on water excess conditions (W/L ≈ 1.3), as applied in sample NRK-2. Under these conditions, quicklime is dispersed in a fluid phase, resulting in the highest BET surface area of the study (35.4 m2/g), together with high pore volume. The main drawback of this approach is the formation of a wet slurry that requires post-drying, with partial recarbonation of portlandite to calcite during this step. This behaviour is consistent with slurry-based patented processes reported in the literature [25], in which enhanced dispersion promotes porosity development. By contrast, pressure-controlled hydration follows a fundamentally different process route, with distinct implications for MgO conversion and sorbent performance.
An alternative approach is based on stoichiometric hydration combined with the addition of a dispersing additive such as triethanolamine (TEA), as applied in sample NRK-3. This method produces a highly reactive hydrated lime with high BET surface area, while maintaining low residual moisture and avoiding the need for post-drying. Compared with NRK-2, additive-assisted hydration results in slightly lower BET surface area but higher portlandite purity and lower residual moisture content.
Two main approaches were identified for producing high-surface-area hydrated limes: excess-water hydration, which maximizes surface area but requires post-drying and may induce recarbonation, and additive-assisted hydration, which yields slightly lower surface area but higher portlandite purity and reduced residual moisture. Together, these approaches highlight a trade-off between porosity development and product stability depending on the targeted application.
5.2.2 Relationship between hydration extent and BET surface area
To further investigate the relationship between hydration extent and porosity development, BET specific surface area was plotted as a function of the total content of hydrated phases (portlandite + brucite) (Figure 7). The resulting trend is distinctly non-linear and characterized by clear threshold behaviour. At low contents of hydrated phases, corresponding mainly to NHL and SCM-derived materials, BET surface area remains limited (<10 m2/g). Conversely, once a critical amount of hydrated phases is reached, a sharp increase in BET surface area is observed, followed by a plateau region (≈20–35 m2/g), where values remain high despite further increases in hydration extent. This behaviour indicates that the formation of hydrated phases is a necessary condition for porosity development but does not directly control its magnitude beyond a critical threshold. Once sufficient portlandite and/or brucite has formed, the final surface area is primarily governed by process-related factors, including water availability, dispersion efficiency, and hydration kinetics. This interpretation helps explain why samples with comparable degrees of hydration, such as NRK-2 and NRK-3, exhibit significantly different BET surface areas, and why SCM-based materials, despite partial hydration, fail to develop significant porosity due to their limited initial CaO content.
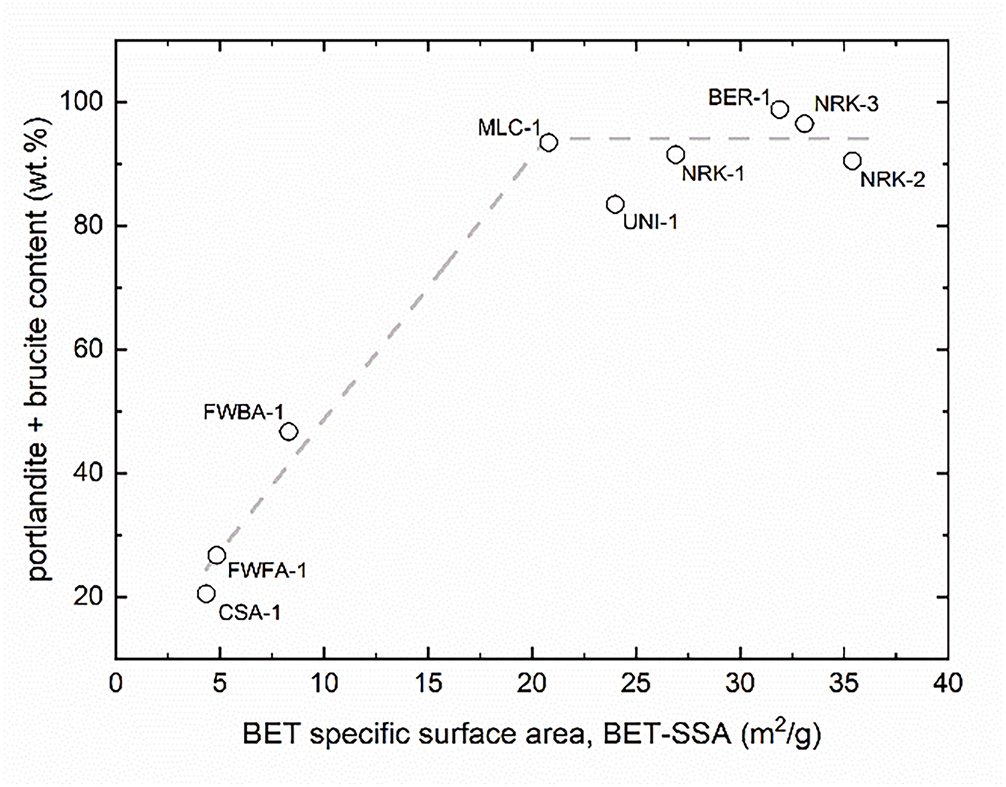
Figure 7 BET specific surface area vs. total hydrated phases (portlandite + brucite), showing a non-linear threshold behaviour with a sharp increase followed by a plateau region
Overall, the development of surface area in hydrated lime systems can be described as a general two-step mechanism: (i) a composition-controlled stage, in which the availability of reactive oxides enables the formation of hydrated phases, and (ii) a process-controlled stage, governed by the hydration pathway and microstructural evolution. This interpretation highlights that achieving high BET surface area requires not only sufficient hydration, but also careful control of process conditions.
5.2.3 Pressure-controlled hydration of dolomitic lime
A third approach involves the hydration of dolomitic limes under pressure-controlled conditions [26] (UNI-1 and BER-1). The process follows a two-step mechanism, with rapid hydration of CaO followed by pressure-assisted hydration of periclase to brucite. Maintaining elevated temperature and controlled pressure is therefore essential to promote MgO hydration, as also highlighted in patented pressure-controlled processes [27].
Under these conditions, double-hydrated dolomitic limes with high BET surface area can be obtained. The BER-1 sample exhibited the best performance, combining high surface area, high pore volume, and nearly complete MgO conversion, making it suitable for FGD applications. By contrast, UNI-1 showed lower porosimetric development and residual periclase, making it less suitable for high-performance sorbent applications, although still compatible with less demanding construction uses.
The lower performance of UNI-1 is likely related to material-related factors, including pre-hydration, the presence of unburnt calcite, reduced reactivity due to over-burning, and coarser particle size. Although additional work is required to quantify the relative contribution of these factors, the results demonstrate that pressure-controlled hydration can produce dolomitic sorbents with performances comparable to those of calcitic hydrated limes. These results confirm that pressure-controlled hydration represents a fundamentally different process route compared to slurry-based systems, with distinct implications for MgO conversion and sorbent performance.
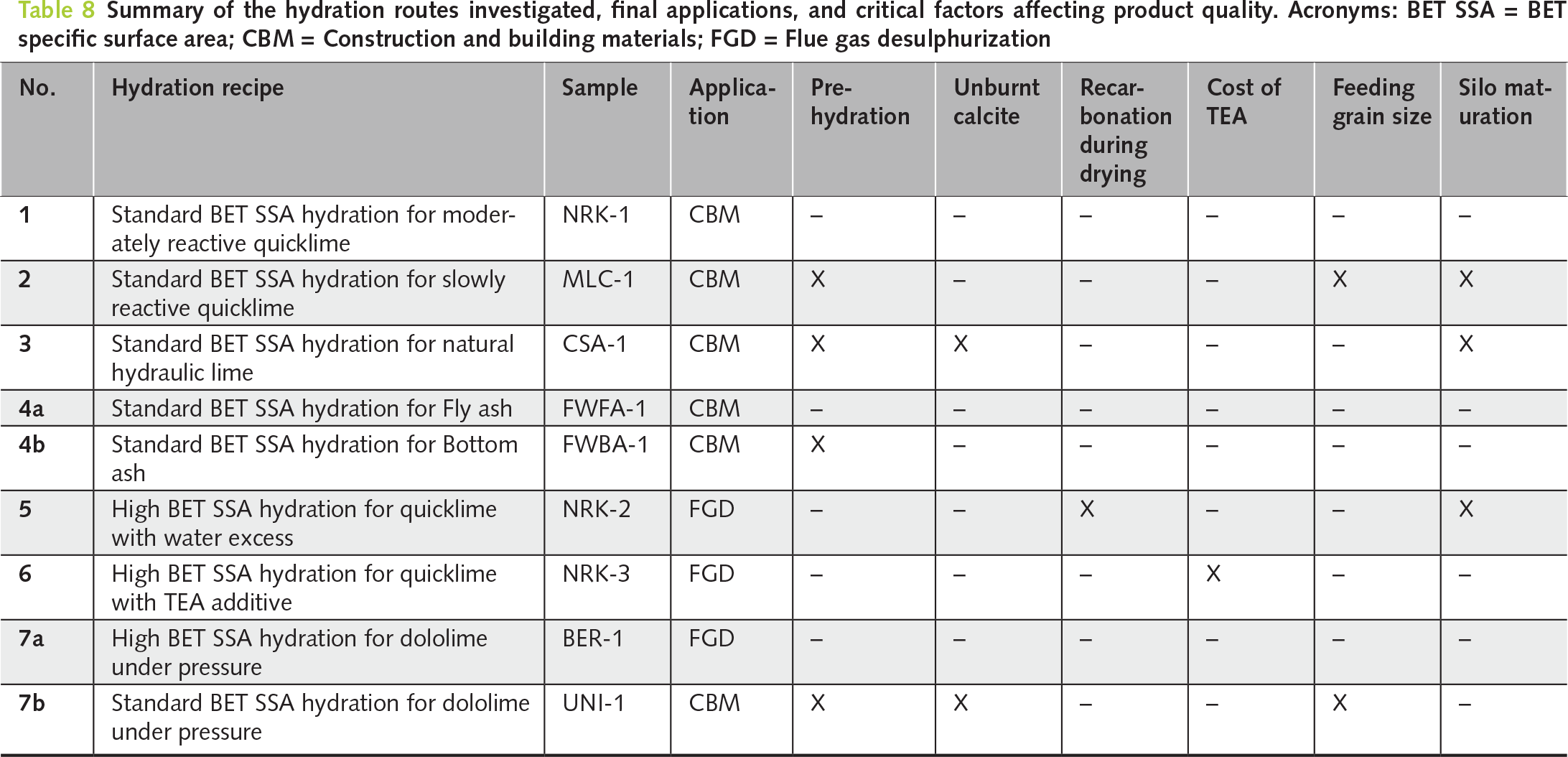
Table 8 summarizes the main hydration routes investigated and highlights the critical factors affecting product quality for each application. From an industrial perspective, the results show that controlled laboratory hydration can support raw material screening, operating window selection, and prediction of product performance before full-scale production. In this sense, the planetary mixer represents a practical tool for reducing scale-up risk and improving industrial process design.
The main findings of this study can be summarized as follows:
1. Validation of the experimental system. The purpose-built fully integrated planetary mixer proved to be an effective tool for simulating industrial lime hydration under controlled laboratory-scale conditions, enabling precise control of temperature, pressure, water dosage, and mixing dynamics.
2. Role of raw materials and process parameters. Hydration behaviour is primarily controlled by raw material properties, particularly available lime content (ALI) and reactivity, as well as by process parameters. Their combined effect governs phase conversion and the development of porosimetric properties.
3. Performance of SCM by-products in hydration processes. The hydration of SCM by-products demonstrated their potential reuse as partially reactive systems in construction materials, supporting circular economy strategies, although their limited CaO content restricts full hydration and porosity development.
4. Mechanism of BET surface area development. The evolution of BET surface area follows a two-step mechanism: (i) a composition-controlled stage related to the availability of reactive oxides, and (ii) a process-controlled stage governed by hydration conditions and microstructural development.
5. Technological routes for high-BET hydrated limes. Two main approaches were identified for producing high-surface area hydrated limes:
• excess-water hydration, which maximizes surface area but requires post-drying and may induce recarbonation.
• Additive-assisted hydration, which yields slightly lower surface area but higher portlandite purity and reduced residual moisture.
These approaches highlight a trade-off between porosity development and product stability depending on the targeted application.
6. Pressure-controlled hydration of dolomitic limes. Pressure-assisted hydration enables efficient MgO conversion to brucite allowing the production of high-performance dolomitic sorbents with properties comparable to those of calcitic hydrated limes.
7. Industrial implications. The proposed approach establishes a direct link between laboratory testing and industrial hydrator operation, supporting raw material screening and reducing scale-up risks. It provides a robust framework for bridging laboratory-scale experimentation and industrial hydrator design, with direct implications for process optimization and material valorization.
[1] Boynton R. Chemistry and technology of lime and limestone. 2nd ed. New York, NY, USA: Wiley; 1980.
[2] Oates JAH. Lime and limestone. Chemistry and technology, production and uses. Weinheim, Germany: Wiley-VCH; 1998.
[3] Elsen JMG, Mertens G, Snellings R. Portland cement and other calcareous hydraulic binders. In: Christidis GE, editor. Advances in the characterization of industrial minerals. EMU notes in mineralogy. 2011. p. 441–79. doi:10.1180/EMU-notes.9.11.
[4] Schorcht F, Kourti I, Scalet B, Roudier S, Sancho LD. Best available techniques (BAT) reference document for the production of cement, lime and magnesium oxide. Luxembourg: JRC; 2013 [cited 2026 April 26]. Available from: https://bureau-industrial-transformation.jrc.ec.europa.eu/sites/default/files/2019-11/CLM_Published_def_0.pdf.
[5] Hai Do DSE, Kehse G, Ferri V, Christiansen T, Bresciani P. Simulation of lime calcination in PFR kiln. Influence of energy input and lime throughput. ZKG Int. 2012;65(12):52–64.
[6] Vola G, Sarandrea L, Della Porta G, Cavallo A, Jadoul F, Cruciani G. The influence of petrography, mineralogy, and chemistry on burnability and reactivity of quicklime produced in Twin Shaft Regenerative (TSR) kilns from Neoarchean limestone (Transvaal Supergroup, South Africa). Miner Petrol. 2018;112(4):555–76. doi:10.1007/s00710-017-0542-y.
[7] Vola G, Bresciani P, Rodeghero E, Sarandrea L, Cruciani G. Impact of rock fabric, thermal behavior, and carbonate decomposition kinetics on quicklime industrial production and slaking reactivity. J Therm Anal Calorim. 2019;136(3):967–93. doi:10.1007/s10973-018-7769-7.
[8] Vola G, Sarandrea L, Mazzieri M, Bresciani P, Ardit M. Reactivity and overburning tendency of quicklime burnt at high temperature. ZKG Int. 2019;72:20–31.
[9] Vola G, Ardit M, Sarandrea L, Brignoli G, Natali C, Cavallo A, et al. Investigation and prediction of sticking tendency, blocks formation and occasional melting of lime at HT (1300°C) by the Overburning Test method. Construct Build Mat. 2021;294(1):123577. doi:10.1016/j.conbuildmat.2021.123577.
[10] Vola G, Ardit M, Frijia G, Di Benedetto F, Fornasier F, Lugli F, et al. Characterization and provenance of carbonate rocks for quicklime and dololime production in twin shaft regenerative kilns from the Arabian Peninsula and neighboring countries. Minerals. 2023;13(12):1500. doi:10.3390/min13121500.
[11] Revuelta M. Lime. In: Construction materials—geology, production and applications. Cham, Switzerland: Springer; 2021. p. 167–93. doi:10.1007/978-3-030-65207-4_7.
[12] Cimprogetti: hydration plants, technical brochure. 2018 [cited 2026 April 26]. Available from: https://www.cimprogetti.com/2018_03_depliant_hydration_EN_EMAIL.pdf.
[13] UNI EN 459-1. Building lime—part 1: definitions, specifications and conformity criteria. Brussels, Belgium: CEN; 2015.
[14] ASTM C206-14. Standard specification for finishing hydrated lime. West Conshohocken, PA, USA: ASTM; 2014.
[15] UNI EN 197-1. Cement—part 1: composition, specifications and conformity criteria for common cements. Brussels, Belgium: CEN; 2011.
[16] Sonebi MAY, Ammar Y, Diederich P. Sustainability of cement, concrete and cement replacement materials in construction. In: Khatib JM, editor. Sustainability of construction materials. Cambridge, UK: Woodhead Publishing; 2016. p. 371–96. doi:10.1016/B978-0-08-100370-1.00015-9.
[17] Martinez CM, Sáez del Bosque IF, Medina G, Frías M, Sánchez de Rojas MI. Fillers and additions from industrial waste for recycled aggregate concrete. In: The structural integrity of recycled aggregate concrete produced with fillers and pozzolans. Cambridge, UK: Woodhead Publishing; 2022. p. 105–43. doi:10.1016/B978-0-12-824105-9.00012-3.
[18] Bish DL, Howard SA. Quantitative phase analysis using the Rietveld method. J Appl Cryst. 1988;21(2):86–91. doi:10.1107/S0021889887009415.
[19] Young R, editors. The rietveld method. Oxford, UK: Oxford University Press; 1993. doi:10.1093/oso/9780198555773.001.0001.
[20] Döbelin N. Profex—a graphical user interface for the Rietveld refinement program BGMN. 2025 [cited 2026 April 26]. Available from: https://www.profex-xrd.org/.
[21] ISO 9277. Determination of the specific surface area of solids by gas adsorption (BET method). Geneva, Switzerland: ISO; 2022.
[22] Micromeritics: an introduction to NLDFT models for porosity characterization, application note. [cited 2026 April 26]. Available from: https://it.scribd.com/document/713209930/WP-An-Introduction-to-NLDFT.
[23] ASTM C25-19. Standard test methods for chemical analysis of limestone, quicklime, and hydrated lime. West Conshohocken, PA, USA: ASTM; 2019.
[24] UNI EN 459-2. Building lime—part 2: test methods. Brussels, Belgium: CEN; 2021.
[25] Chini S, Lorgouilloux M, Nyssen O, Francoisse O. Process for manufacturing highly porous slaked lime, EP3858799A1. 2021 [cited 2026 April 26]. Available from: https://patentimages.storage.googleapis.com/73/b2/c8/b3b32838b36e89/EP3858799A1.pdf.
[26] Massa M, Vola G, Bresciani P, Colombari V, Bocchialini G, Brazzo P, et al. Insights into the mild-hydrothermal hydration mechanisms and material properties of dololimes and Mg-lime. Powder Technol. 2026;479(11):122568. doi:10.1016/j.powtec.2026.122568.
[27] Corson B. Process for producing dry lime hydrate. United States patent US 3,106,453. 1963 Oct 8 [cited 2026 April 26]. Available from: https://patentimages.storage.googleapis.com/0e/59/20/c3a0903f9664fd/US3106453.pdf.