Influence of material properties on the decomposition time of limestone under shaft kiln conditions
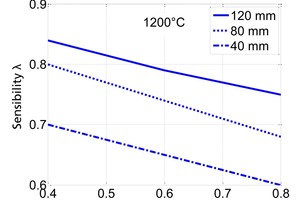
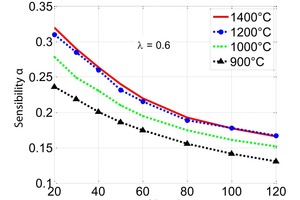
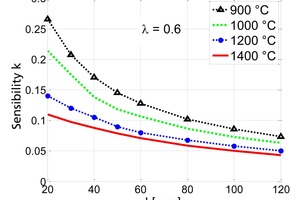
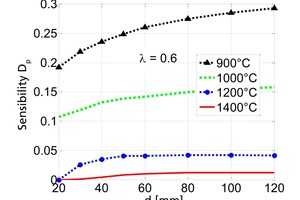
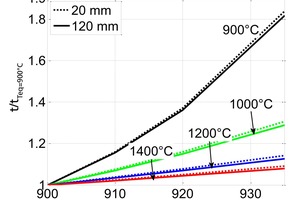
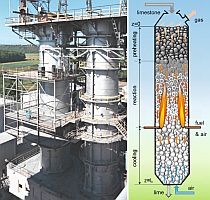
The influence of the material properties on the decomposition of limestone particles under shaft kiln conditions has been investigated. Limestone particles are assumed to be spherical particles and the shrinking core model has been applied. In counter-flow single-shaft kilns with high temperatures, the thermal conductivity of the lime has the dominant influence with up to 90 % for large particles. In parallel-flow regenerative (PFR) shaft kilns with lower temperatures, the reaction coefficient and the equilibrium pressure also have a significant influence in addition to the thermal conductivity. These three material properties can fluctuate up to a factor of two depending on the origin of the limestone.
1 Introduction
The decomposition of limestone is an endothermic topochemical reaction:
CaCO3 + ΔHR = CaO + CO2
(solid) (reaction enthalpy) (solid) (gaseous)
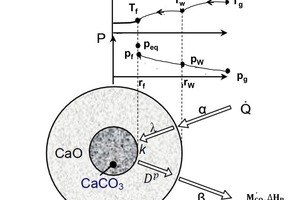
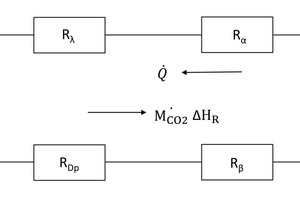
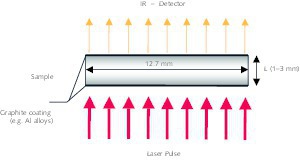
The calcination process can be explained by using a partially decomposed piece of carbonate [1, 2]. The calcination profiles of CO2 partial pressure and temperature are shown in Figure 1. The specimen comprises a dense carbonate core surrounded by a porous lime layer. In the kiln the combustion gas with the temperature Tg transfers heat by convection (symbolized by α) to the solid surface with the...