Brick powders as pozzolanic additives in cement production
In a research project, 26 different brick powders made from ground bricks of different ages and different product ranges were tested with regard to their suitability for use as pozzolanic components of cement or as an additive to concrete. It was found that the use of brick powder as a ”supplementary cementitious material“ is not only possible, but also worthwhile. Different approaches are conceivable when producing suitable brick powders.
1 Introduction
The shift away from fossil fuels, which are still the primary energy source for heat and power generation, has already been decided. This will result in radical changes in the secondary raw materials industry. In the building materials sector, this will impact i.a. the use of hard coal fly ash, which will then no longer be available as a composite in cement manufacture or as an additive in concrete production. Compensating for this loss by increasing the clinker content in the cement would be counterproductive because it would increase the CO2 emissions of the cement industry. Instead, new additives have to be found. Research in this sector focuses on materials such as limestone, burnt clays and natural pozzolans. In addition, various residues from ore and natural stone processing, dredged material, slags and incineration ashes from organic materials are being tested.
One rather neglected area of material recycling is the recovery of construction waste – especially masonry rubble. These mixtures of brick, mineral wall-building materials and mortar and plaster occur in Germany in quantities of approx. 25 million t/a [1]. The brick content of around 10 million t or even the entire mixture could be used as an alternative raw material for the production of a pozzolana additive. This would compensate for changes in the material flows caused by the energy transition and at the same time improve the level of recovery for a proportion of the occurring construction waste.
2 State of knowledge
A large number of studies are available on the use of brick powder as an additive in cement production. In many cases these were designed in such a way that only a selected brick powder was added to the cement in different proportions. The focus of the studies was the change in strength of the mortars and concretes thereby produced.
For the summary of the results (Figure 1), the determined strengths were related to the strength of the original cement and shown as a function of the substituted cement content. This result showed a decrease in strength in line with an increasing proportion of brick powder. The majority of the measured values lie above the assumed linear dilution curve. This means that a filler effect can be assumed, which has a positive influence on the strength. In the case of substituted proportions of up to 15 %, some powders contribute to the strength formation.
The reasons for the different effects on the strength can not yet be clearly stated. Influences originating from the raw materials used, the type of brick and its respective manufacturing conditions – the firing temperature can range between 900 °C for masonry bricks and 1200 °C for clinker bricks – were not investigated. A number of authors investigated the influence of the fineness of the powders. According to the study by Irki [15] a 20 % substitution of cement with brick powder increased the 28-day strength from 38.3 MPa at a specific surface of 3900 cm²/g through 40.4 MPa at 4300 cm²/g to 46.0 MPa at 5200 cm²/g. In contrast, Zhao [20] was unable to register any increase in strength with a 30 % substitution and an increase in the specific surface from 2533 cm²/g to 7954 cm²/g.
Rahhel [18] describes the crystalline phases formed during the hydration of mixtures comprising 40 % brick powder and 60 % Portland cement. After two days of hydration, ettringite and portlandite are found as hydration products that can be detected by X-ray analysis. As from day 7, these are joined by hemicarboaluminate C4AC—0.5H11.5 and as from day 28 by monocarbo-aluminate C4AC—xH11. These phases can also be detected during the hydration of calcined clays [21]. This suggests a cross-connection between bricks and calcined clays. O’Farell therefore investigated both the clays fired at 800 °C and the bricks technically manufactured from these clays [22]. His tests showed (Figure 4) that the bricks also exceeded the dilution curve, but by significantly less than the calcined clays. The cements containing brick powder achieved somewhat lower strengths than the cements containing the fired clays.
Severins presents distinctly more practice-oriented test results [23]. These results show that cements of strength class 42.5 can be produced with up to 30 % addition by mass of powder from ground masonry rubble. Based on the findings regarding the resistance to carbonation, frost and freeze-thaw cycling, as well as against the penetration of chloride ions, Severins concluded that these cements are suitable for the production of concrete components for structural engineering and industrial construction.
Despite the diversity of the results, the litera-ture evaluation shows that it is worthwhile to continue investigating the use of brick powder as “supplementary cementitious materials”. In order to take into account the influences of the primary products, the suitability of various types of brick as a pozzolanic additive was tested. The intention was to prepare the use of brick powder obtained from masonry rubble as an additive. Parallel to this, it is important to develop and launch technology concepts for processing masonry rubble into homogeneous brick powders.
3 Material and methods
The investigations included 14 new and 12 used bricks from different product ranges. The used bricks were selected or pretreated in such a way that no mortar or plaster was adhering to them. In the case of thermal insulating bricks, the insulation material was removed before processing. Two delivery batches of Portland cement CEM I 42.5 were used as binders.
To characterize the starting material, the absolute powder density and the oven dry particle density were determined using helium comparison pycnometry resp. powder pycnometry. The chemical composition was determined with the aid of X-ray fluorescence analysis and the loss on ignition was determined in accordance with DIN 51081. X-ray diffractometry was used to determine the mineralogical composition.
After pre-crushing with a jaw crusher, the brick samples were ground in a ball mill to particle sizes < 63 µm. To determine the influence of fineness on the reactivity and strength, 15 of the 26 samples were ground again. The particle size distribution of the powders was determined using laser granulometry.
The powder-fine samples were used for investigating the reactivity and for determining the strength. The Chapelle test and the simultaneous thermal analysis (STA) were used for determining the reactivity. With the Chapelle test, the consumption of calcium hydroxide as a result of the chemical reactions with silicon and aluminum-rich reactive substitute components is recorded in accordance with the French standard NF P 18-513:2010 [24]. 250 ml of distilled water was added to a weighted sample of 3 g of brick powder and 2 g of CaO then was mixed in a sealed vessel using a magnetic stirrer. The Ca(OH)2 consumption was determined by titration with 0.1 molar hydrochloric acid. However, differing from the standard, this was done only after a storage period of 28 or 90 days at room temperature. For the simultaneous thermal analysis, pastes were made from 60 % by mass of Portland cement and 40 % by mass of brick powder with a water to binder ratio of 0.7. The hydration was stopped after 28 and 90 days by grinding, adding isopropanol and drying (< 40 °C). The sample prepared in this way was then analysed using the STA.
The strength tests were carried out on standard mortar prisms made of 450 g of binder, 1350 g of standard sand and 225 g of water. The binder consisted of 25 % by mass of brick powder and 75 % by mass of Portland cement CEM I 42.5. Mortar with 100 % Portland cement served as a reference. As two delivery batches of Portland cement were used in the course of the project, the related compressive strength had to be used as an evaluation standard.
4 Characterization of the starting material
The bricks used as the starting material had absolute powder densities ranging from 2.64 g/cm³ to 2.79 g/cm³, particle densities (OD) ranging from 1.39 g/cm³ to 2.40 g/cm³ and calculated body porosities of between 12 % by volume and 50 % by volume. The increase in true density is due to shifts in the mineralogical composition from quartz to heavier mineral phases such as feldspars and hematite. The densities of the clinker bricks were above 2.0 g/cm³. The tested masonry bricks and roof tiles had densities of 1.51 g/cm³ to 2.05 g/cm³. In the case of thermal insulating bricks, values of around 1.4 g/cm² could be achieved by adding pore forming materials.
Table 1 provides an overview of the chemical composition of the raw materials used. This shows that there were very SiO2-rich “acidic” and relatively CaO-rich “basic” samples. The SiO2-rich samples include the clinker and some roof tiles and masonry bricks. The thermal insulating bricks and some masonry bricks represent the CaO-rich samples.
The ternary system SiO2-Al2O3-CaO can be used to assess the suitability in principle of “supplementary cementitious materials”. Based on the entered compositions of the 26 bricks, 11 brick varieties were found to lie above the range designated for natural pozzolans. These included two roof tiles and two masonry bricks as well as all clinkers. Five samples were located in the range of fly ashes. 10 samples could not be assigned to either range.
The mineralogical analysis showed quartz, feldspar, hematite and amorphous phases. In the thermal insulating bricks, residues of clay minerals were found, which is due to the low firing temperature of this type of brick.
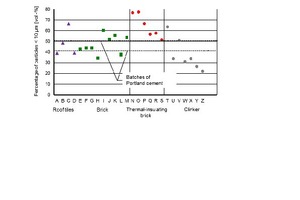
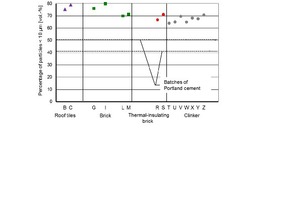
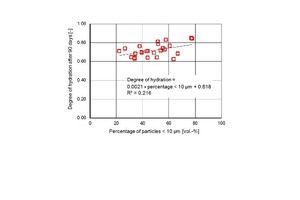
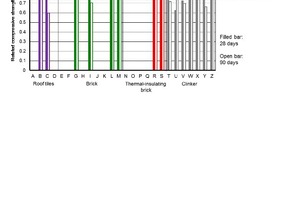
In order to evaluate the fineness of the brick powders, the proportion of particles <10 µm from the measured distributions was used. The idea behind this approach was that only fine particles would have an effect on strength development by improving the packing density and/or participating in hydration. The comparison in Figure 5 shows that there are considerable differences. The clinker Z with a fine particle content of 20.7 % by volume was the coarsest material and the thermal insulating brick O with a content of 75.8 % by volume was the finest material. The greater number of the brick samples had smaller portions of the faction < 10 µm than the cements. After the second grinding there were more uniform fine fractions, which were significantly higher than those of the cements (Figure 6).
The differences in the fineness of grinding after the first grinding correspond to the different crushing resistances of the bricks. These are in turn influenced by the porosity and the SiO2 content, which represents the quartz content. The clinker with low porosity and high SiO2 content as well as some roof tiles and masonry bricks have higher crushing resistances than the thermal insulating bricks. As a result, the powders produced from them had a lower proportion of particles <10 µm after the first grinding.
5 Reactivity and strength
The consumption of Ca(OH)2 after 90 days measured with the Chapelle test was between a minimum of 97 mg/g and a maximum of 328 mg/g (Figure 7). These values are in the same order of magnitude as the calcined clays when these are measured like the brick powders with the modified version of the Chapelle test. There is a tendency that the Ca(OH)2 consumption increases in line with an increasing proportion of particles <10 µm – that is, with increasing fineness. All the Chapelle values of the reground powders are higher than those of the first grinding, with the exception of the values of the thermal insulating bricks. The large scatter indicates that, in addition to the fineness of the powders, there are other influences, such as the CaO or SiO2 content, which can be used to distinguish the types of bricks. In order to investigate these influences, DTA analyses were carried out on the samples from the first grinding and used for determining the progress of hydration on the basis of the following para-meters:
The content of LOI-resistant content as measure of the non-hydrated components, which corresponds to the residual mass at the end of the DTA measurement if the fed sample material is pre-dried at 40 °C
The degree of hydration as measure of the progress of the reaction, which follows from the water bound in the calcium and aluminate silicate hydrates and in the portlandite, taking into account the partial conversion of the portlandite to carbonate based on the sample masses at staggered temperatures [7]
Degree of hydration =⇥(1)
W105; W600; W1000: Sample masses at 105, 600, 100 °C
n = 0.24: Chemically bound water in the completely hydrated samples
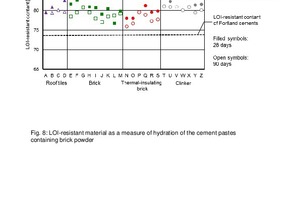
Referred to the initial value of 100 % by mass for the unhydrated samples, the LOI-resistant content for the pastes containing brick powder decreases to 80 to 85 % by mass after a hydration period of 28 days (Figure 8).
With increasing duration, the content continues to decrease due to the progressive hydrate phase formation and reaches values below 80 % by mass after 90 days. There is a tendency for hydration of such brick samples, which are initially very sluggish, to accelerate with increasing hydration time. The LOI-resistant content of the pastes made from the Portland cement has already reached its final value after 28 days with 74 % by mass. The Portland cement is therefore completely hydrated. A mixture of 60 % cement and 40 % brick powder without hydration involvement should have 0.6 x 74.0 + 0.4 x 100 = 84.4 % LOI-resistant content, which is also the case for some brick samples after a hydration period of 28 days.
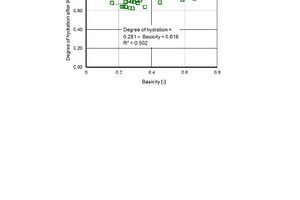
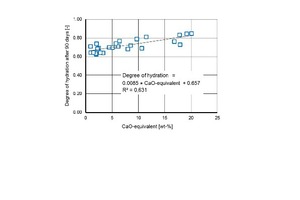
The degree of hydration ranges between 55 % and 77 % after 28 days and between 63 % and 85 % after 90 days. It is influenced with less significance by the fineness of the brick powders than by the type of brick (Figures 9, 10 and 11). Basicity [26] or the CaO equivalent are good ways of describing the type of brick as an influencing variable with a numerical value:
Basicity = (CaO + MgO + Al2O3) ⇥(2)
SiO2
⇥
CaO-equivalent = CaO + 1.39 ∙ MgO⇥(3)
6 Strength
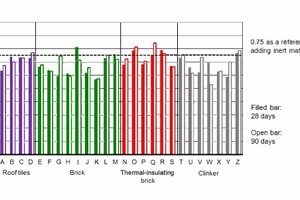
The strength of the mortar made with the cement containing brick powder (Figure 12) shows great differences after 28 and 90 days, respectively. The reference value, which results from replacing 25 % of Portland cement with inert rock powder, is exceeded after 28 days by 6 and after 90 days by 8 out of the total of 26 mortars. With a few exceptions, strength increased between the 28th and 90th day. The repeated grinding of 15 selected brick samples caused an increase in strength in almost all samples, so that the masonry bricks and the clinker now exceeded the reference value of 0.75 for the addition of inert powder (Figure 13).
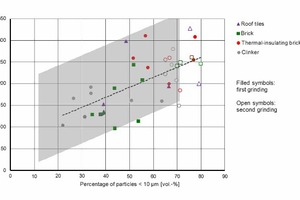
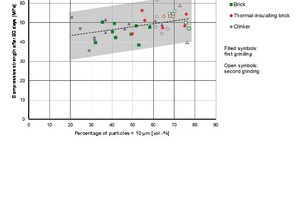
This fact can also be seen in the dependency between the fineness of the powders and the 90-day strength in Figure 14. If the proportion of particles <10 µm increases due to finer grinding, there is an increase in strength, particularly in the case of clinker powders. The differences between the various types of bricks decrease in line with the increasing proportion of fine powder.
7 Summary and conclusions
In a research project, 26 powders made from bricks of different ages and different product ranges were tested with regard to their suitability as pozzolanic components of cement or as an additive to concrete. Based on the chemical composition, it was possible to differentiate between CaO-rich, basic brick types such as thermal insulating bricks and masonry bricks from current production and SiO2-rich, acidic brick types such as clinker bricks. The reactivities determined with a modified Chapelle test were in a range between 97 mg/g and 328 mg/g. At least in part, this lies in the same range as calcined clays. The degrees of hydration, based on DTA analyses of the mixtures comprising 60 % by mass of cement and 40 % by mass of brick powder, reached a maximum of 77 % after 28 days and 85 % after 90 days. Portland cement is 97 % hydrated after just 28 days. The degree of hydration increases with the fineness of the brick powders. The influence of the type of brick can be determined using the CaO and MgO contents combined to form the CaO equivalent. Bricks with high levels of these oxides have the highest degrees of hydration. The brick powders participate to a varying extent in the formation of strength. CaO-rich varieties such as thermal insulating bricks are particularly effective. The SiO2-rich brick types also contribute to the formation of strength if they are ground to a sufficient degree of fineness.
According to the results presented, it seems promising to continue researching this recycling option for bricks from building demolition. Further studies on the spatial stability and the durability, such as frost resistance, carbonation behavior and chloride penetration resistance are required to prepare enable the application of brick powder in concrete.
Different approaches are conceivable for the production of suitable brick powders:
On the one hand, unmixed bricks can be obtained through selective demolition and processed into brick powder as a pozzolanic additive. However, it is questionable whether sufficient quantities of the required quality can be generated with this method
On the other hand, it is possible to make use of masonry rubble, which usually consists of various wall building materials as well as mortar and plaster, as a secondary source of raw materials. Sorting is then required to obtain a homogenous supply of bricks. Conventional density-based sorting methods are not very suitable for this, as the wall-building materials hardly differ in their density [27]. Sensor-based processes are conceivable, but they can only be used above grain sizes of approximately 8 mm, so that it would be possible to utilize only part of the masonry rubble
A third possibility is separation of the brick particles from fine-grained masonry rubble or powder. First results are available regarding the feasibility of this method [27, 28]. The entire brick portion in the masonry rubble could be utilized
The recovery of brick fractions that are as homogenous as possible must be followed by the grinding process. This is the most important technical measure for achieving the necessary reactivity.
//www.iab-weimar.de" target="_blank" >www.iab-weimar.de:www.iab-weimar.de
solution. Cement and Concrete Research 29 (1999) 1781-1790
Cement & Concrete Composites. 23 (2001) 81-91
cementitious material. Cement & Concrete Composites 34 (2012) 55-61
nano-SiO2 particles. Construction and Building Materials 38 (2013) 255-264
resistance of concrete. Construction and Building Materials 55 (2014) 440-446
of Scientific & Engineering Research, Volume 6, Issue 12 (2015) 152-157
low heat of hydration cements. Materials and Structures 51 (2018) 101, 1-12
Sustainability 2018, 10, 1037, 1-16
of blended cement. Journal of Cleaner Production 242 (2020) 118521
Dissertation. Universität der Bundeswehr. 2017
Zementherstellung. 20. Internationale Baustofftagung 12. – 14. September 2018, Weimar
Metakaolin – Definitions, specifications and conformity criteria, 2010
41 (2011) 1244-1256

![1 Related compressive strength of blended cement according to literature [2–20]](/uploads/images/2020/w300_h200_x297_y421_Materials_Mueller_Brick_powders_Figure_1-095a22aaecd10a0c.jpeg)
![2 Comparison of related compressive strength of blended cement with ground waste clay and the bricks produced from it [22]](/uploads/images/2020/w300_h200_x297_y421_Materials_Mueller_Brick_powders_Figure_2-7ef75181d73e3328.jpeg)




![4 Ternary phase diagram SiO2-CaO-Al2O3 with ranges of fly ashes and pozzolana according to [25] compared to the composition of brick powder (see Table 1)](/uploads/images/2020/w300_h200_x297_y421_Materials_Mueller_Brick_powders_Figure_4-62e96c06e7d05758.jpeg)