Durability enhancement for cementitious materials through waterproofing by
organosilicon chemistry
Damage associated with water inside the mass are a key threat to a more durable cementitious product. With the help of organosilicon waterproofing agents, such damage can be very effectively suppressed or fully prevented. In addition to the well-established surface treatment approach, integral waterproofing by organosilicon agents further enables more thorough, multi-faceted protection for cementitious systems in the long run.
1 Introduction
Cementitious materials like concrete are mainly composed of aggregates of various sizes such as sand and rock, which are bonded together by fresh cement paste and become a rigid mass over time. Although mostly hard and dense, cement-based systems are by their very nature still porous and thus susceptible to damage and deterioration caused by deleterious substances. Typical examples of such deterioration processes are alkali-silica reaction, sulphate attack, carbonation, frost damage etc. A common feature generally behind all these processes is the presence of water, in its liquid, solid or gas state. Water can either act as a transport medium for the deleterious substances or be directly involved in the chemical or physical processes [1]. As a result, most construction projects require a certain type of proofing system to make cementitious products impervious to external water, as well as other substances.
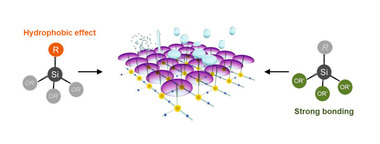
For decades, organosilicon-based chemicals have been used in the construction industry for the protection of various building materials including cementitious types against moisture and consequential deteriorations [2, 3, 4, 5]. For instance, silanes carrying reactive and inert groups have the capability to create a pronounced hydrophobic effect on finished mineral-based substrates after treatment [6]. When they are applied to the substrate material, the reactive groups such as alkoxy silyl groups can react with hydroxyl groups existing on the surface to form stable covalent bonds. The silanes themselves can also react with each other through hydrolysis and condensation processes between the remaining alkoxy silyl groups and can form a very thin but permanent layer of polymer network covering the substrate surface. In contrast, the non-polar groups as the inert part of the silane molecules, e.g. alkyl groups, render the substrate surface hydrophobic. As a result, water or moisture is repelled and cannot penetrate into the substrate structure. With this, water-associated processes such as blistering, fungi growth, salt attack etc., which may easily cause health issues and safety damage to the materials and structures in use, can be significantly suppressed.
Wacker has been a technology leader in the field of organosilicon-based construction chemicals for post concrete surface treatment for more than six decades. The product series which is commercially available under the trade name SILRES® BS has been very effectively applied to treat and protect a wide range of buildings and public works [5, 6, 8]. By means of surface impregnation, coating, etc., these products help achieve excellent hydrophobic effects on treated substrates against water-induced damage. Besides hydrophobicity, silane and siloxane chemistry also offers additional benefits such as anti-stain effects or brilliant wall colors, contributing to the overall aesthetics of construction works.
The application of organosiloxane agents can generate pronounced hydrophobicity within the top layer of the surfaces of walls, floors or other building products. This provides exterior protection against water-associated damage to a very high level. However, there are several occasions where the surface protection alone is insufficient or inappropriate. For instance, if the protective surface layer gets damaged during service, the unprotected inner part will be subject to possible deleterious effects from the surrounding environment. Even with a tiny failed spot on the surface, the subsequent capillary effect can draw moisture from the surrounding environment into the dry mass, leading to water-induced internal deteriorations. Besides, the surfaces of the treated objects need to be dry so that the penetrating and hydrophobizing processes can take place. This means that materials with wet surfaces are not suitable for direct surface treatment. In the case of freshly-mixed concrete, a sufficient drying time of several days or weeks needs to be ensured before hydrophobic agents can be applied.
In order to provide longstanding comprehensive protection, it is also necessary to hydrophobize the interior of the target object and convert the whole of it into a water repellent mass. This is especially critical for cement-based materials, as water is always present inside the cementitious matrix – either as a chemical constituent of the hydration process or from external sources. Moreover, the porous and hydrophilic nature of the matrix renders it susceptible to the deteriorations resulting from transport processes of water, thus impairing the durability of the material significantly.
In this article, the waterproof treatment of a cementitious mass by using organosilicon chemicals will be elaborated. We will be able to show that integral hydrophobation is key to the effective and economical protection against any water-associated deterioration process. It is therefore the ideal approach to enhance the internal durability and reliability of a cementitious system.
2 Integral waterproofing of cementitious materials
2.1 Overview of current approaches
Integral waterproofing is a highly efficient solution to make cementitious materials such as concrete water-repellent. It also has noticeable advantages. For instance, in construction works such as deep underground or tunnelling projects, the dosing of a hydrophobic additive can be easily incorporated into the concrete batching process, just as convenient as any other conventional admixtures. No extra effort is thus needed for achieving the intended waterproofing effect. It can also be very convenient for waterproofing concrete parts of complex structures or blind side work where traditional constructing methods would be difficult to implement and where lots of auxiliary set-ups need to be in place. Furthermore, long-lasting waterproofing will reduce maintenance costs for the whole lifecycle of the concrete work dramatically.
Integral waterproofing can be achieved by using chemicals as hydrophobic admixtures mixed with other constituents [9]. Carbon-based organics such as fatty acid salts have been broadly used to make concrete hydrophobic for many years [10]. The molecules of these chemicals, deposited in the pores and voids within the concrete, form a thin layer covering the inner pore surfaces. Subsequently, such layers can prevent water from penetrating into the pore walls since the alkyl or alkenyl groups of the molecules facing outward are non-polar and hydrophobic by nature. The groups of the inward-facing molecules form a loose bonding with the minerals of the inner pores. However, the bonding is not permanent and is subject to break down over time, as physical or chemical deteriorations occur.
Organosilicon-based chemicals can also be used as hydrophobic admixtures for concrete. Similar to carbon-based chemicals, the molecules of silane or siloxane can also line the capillary pore walls via siloxane linkages, as illustrated in Figure 1.
The alkyl groups of the molecules then render the cementitious matrix hydrophobic. But unlike fatty acid salts’ loose bonding with the inner surfaces of concrete pores, the hydroxyl groups of silanes and siloxanes can chemically react with the hydroxyl groups on the concrete pore walls. Through a condensation process, strong -Si-O- bonds are formed and thus silanes and siloxanes are firmly attached to the pore walls. Such condensation reaction can also take place between neighboring silanes and siloxanes to form a polysiloxane network, if residual hydroxyl groups exist. Consequently, the polysiloxane coverage becomes an integral part of the concrete pore system, leading to greatly enhanced durability performance (see Figure 2, right) in contrast to that of an oil-based admixture which is only deposited within the capillaries (see Figure 2,
left).
Apart from hydrophobic admixtures, there is another type typically known as crystalline technology. These admixtures are essentially composed of very fine powder-like ‘reactive’ particles which can function as pore-fillers by hydrating with freely available water inside the pores of the concrete mass [11]. Such reactivity originates from their latent hydraulicity or pozzolanic activity, which can also help to enhance the calcium-silicate-hydrate (C-S-H) gel formation of cement hydration. Inside hardened concrete, the hydration products of these reactive substances can physically block the pores or possible passages to the pores. Consequently, the concrete matrix is converted into a water-resistant state, blocking its pores effectively against any flow of liquids, even under substantial hydrostatic pressure.
2.2 Concrete hydrophobization with organosilicon admixtures
Experimental studies have been conducted by Wacker to compare the waterproofing effect of organosilicon-based agents with conventional additives. The selected admixtures include a silane/siloxane emulsion, a silane and an oil-based powder, and a powder agent based on crystalline technology. The mix designs of the respective test groups are shown in Table 1. The mixing procedures of the test groups were conducted in accordance with BS EN 480-1:2014. As a prerequisite for the sample preparation, the slump value of each mixture was controlled to be within 130-150 mm, by fine-tuning the respective dosage of the superplasticizer.
The basic fresh and hardened properties of the concrete specimens are shown in Table 2. The resistance to water penetration was measured with the specimens completely immersed in water, as shown in Figure 3. It is evident that all hydrophobic agents contribute to decreased capillary water absorption, when compared to the untreated control specimen. The results thus clearly indicate that the tested organosilicon chemicals are effective as concrete admixture in reducing the water absorption of the concrete specimens. Moreover, the water-repelling performance of the mixture containing a 0.4% siloxane emulsion dosed by weight of cement (bwoc) and the 0.2% silane powder mix are on par with the 1% oil-based hydrophobic agent. In order to achieve a similar water-repellent effect, the dosage of the siloxane-based agents can be significantly lower than that of the conventional oil-based agent. This is a strong indication that organosilicon-based products are more efficient when it comes to hydrophobic performance. There are basically two reasons for this: first, siloxane molecules cover the capillary pores more effectively than other agents, and second, the strong bonding between the siloxanes and the minerals of the concrete helps strongly maintain such an effect.
The result also shows that the crystalline product absorbs higher amounts of water than the control specimen. This indicates that under the corresponding test scheme, the crystalline product produced a water-absorbing rather than a water-repellent effect. This is in line with the working principles of the crystalline technology, as it aims at forming, in the first place, pore blocking hydrates in the concrete matrix with the initial water ingress.
In addition to testing the water absorption, the concrete specimens admixed with siloxane and crystalline agents were also subject to a 72 hours water penetration test under a hydraulic pressure of 5 bar. Although the crystalline agent specimen performs slightly better, the results in Figure 4 also show a noticeably lower penetration depth of the specimen treated with a 0.4% dose of Silres BS 1803 when compared to the control specimen. However, it is important to note that siloxanes do not physically block any pores in the inner part of the capillary system. These pores remain open and fully intact. Waterproofing under hydraulic pressure is merely achieved by modifying the wettability of the capillary pore system.
The sample treated with a 0.2% dose of Silres Powder E shows no significant change of penetration depth as compared to the control group. This could be due to its rather low dosage level as compared to the other specimens. Its hydrophobic modification of the capillary pores was not sufficient to markedly resist water penetration under pressure, as compared to Silres BS 1803. Regarding the penetration depth, the crystalline group produced results better than the others. This is no surprise, since crystalline agents have a different working principle. They physically block the pores in the substrate, making them less susceptible to water penetration under pressure.
2.3 Cement hydrophobization with organosilicon additives
Beyond the treatment of concrete, it is possible to hydrophobize the cement itself. Organosilicon additives are able to make cement hydrophobic by covering cement grains with thin layers of hydrophobic organosilicons. An example is shown in Figure 5. Water droplets were placed on treated cement powder (left) and on an untreated sample (right). The water droplet on the untreated cement immediately seeped in through open channels and wetted most of the powder on its way downwards. The water droplet on the left remained on the surface of the hydrophobized cement and was unable to penetrate the powder beneath. Cement treated with organosilicon hydrophobic additives can even repel water while being static and is not susceptible to any surrounding moisture. Organosilicon-based hydrophobic agents are therefore able to:
help stabilize cement against prehydration under humid and warm conditions, especially when the cement is stored in bags.
improve the anti-caking property of cement, as the moisture-reliant agglomeration tendency of cement particles is suppressed by the introduced hydrophobicity.
manufacture hydrophobic cement which can potentially function as a carrier for incorporating hydrophobic properties to cementitious mixtures, e.g. concrete, mortar etc.
3 Mitigation of alkali-silica-reaction in concrete
3.1 Overview of current approaches
Alkali-silica-reaction (ASR) is a well-known durability issue threatening the long-term use of concrete. It can take place when raw materials containing an adequate quantity of alkali and reactive silica are used to produce a concrete. The effective alkali and reactive silica are able to react with each other in the pore solution environment in the concrete to form an alkali-silica gel. The gel product swells if free moisture is available and creates a volume increase that builds up stress inside the rigid mass. This can cause significant weakening of the cementitious matrix and lead to gradual functional failure of the concrete.
Improper raw materials (mainly aggregates) due to resource scarcity or simply operational restrictions are additional reasons why ASR issues occur nowadays. If left unattended or not properly dealt with, the mechanical and durability damage can be substantial. Although rarely with immediate failure, damaged concrete structures or units do need to be repaired or replaced eventually to restore full functionality. Preventive measures to help deal with the potential ASR risk do exist. For instance, sourcing raw materials with low effective alkali and reactive silica contents is one option. Using ASR-inhibiting admixtures such as compounds based on lithium is another [12]. However, these measures are not always feasible. Suitable raw materials can be scarce and therefore too expensive to source in the affected regions. Moreover, transporting large amounts of aggregates over long distance imposes high environmental impact and is not ecological in the long run, either.
3.2 ASR mitigation with organosilicon admixtures
As shown in the previous section, organosilicon hydrophobic admixtures are able to effectively hydrophobize a cementitious mass. Since the damaging processes of ASR need water to be present, hydrophobic treatment should be able to significantly reduce or even completely inhibit the destructive processes. Experimental investigations have thus been carried out by Wacker to examine the corresponding influences. Two types of admixtures were tested: one admixture was silane-based, the other was a silane/siloxane emulsion. Two types of ASR-prone aggregates were used to drastically increase the possibility of the ASR effect in the concrete specimens. The mix designs of respective test groups listed in Table 3 are all based on the top-layer concrete composition suggested in the General Circular on Road Construction (ARS) No. 04/2013, issued by the Federal Highway Research Institute of Germany (BASt) [13].
In order to evaluate the ASR resistance, a testing method developed by the Association of German Cement Manufacturers (VDZ) was adopted [14]. According to this method, concrete prisms with certain dimensions were subject to cyclic exposures to 3% and 10% NaCl solutions which act as external alkali supply. To pass the ASR test, the volume expansion of the specimens is not allowed to exceed 0.30 mm/m with 3% NaCl solution and/or 0.50 mm/m with 10% NaCl solution, after 10 cycles.
The results of the ASR tests with graywacke aggregates, up to 14 exposure cycles with a 3% NaCl solution, are shown in Figure 6, left. The average strain of the control specimen increased steadily over time, and after 10 exposure cycles to the NaCl solution, it measured 0.39 mm/m, exceeding the expansion limit of 0.3 mm/m. This is consistent with the fact that graywacke aggregates are very ASR-sensitive and can greatly enhance ASR effects if they are used in ordinary concrete recipes. However, the specimens containing the silane admix and silane/siloxane emulsion were not affected at all. The average strain values during the overall testing period varied between 0.04 and 0.05 mm/m, respectively. This clearly shows that both the silane agent as well as the silane/siloxane emulsion can significantly inhibit ASR-induced expansion with 3% NaCl solution as alkali supply.
In the second test, rhyolite aggregates were exposed to the 3% NaCl solution up to 14 times, as shown in Figure 6, right. Compared to the graywacke group, the average strain increase of the control specimen was much lower. The strain value amounted to 0.24 mm/m after 10 cycles and did not exceed the 0.3 mm/m limit. This is understandable, since rhyolite is less reactive than graywacke with respect to ASR. However, the specimens containing the silane and the silane/siloxane emulsion agents exhibited almost no volume expansion at all. The average strain values detected after 10 cycles varied between 0.04 and -0.04 mm/m, respectively.
The results of the ASR tests using graywacke aggregates exposed to a 10% NaCl solution are shown in Figure 7, left. The strain increase of the control specimens was significant over the whole testing period. After 10 cycles of exposure to the alkali source, the average strain value greatly exceeded the expansion limit of 0.5 mm/m, and reached 1.97 mm/m. It is quite evident therefore that graywacke aggregates can induce an ASR effect in ordinary concrete recipes if repeatedly exposed to a concentrated external alkali supply. The specimens mixed with a silane agent showed a moderate increase of strain over time, but at a rate lower than that of the control group. After 10 exposure cycles, the average strain value reached 0.94 mm/m, which is well above the expansion limit. However, the specimens mixed with the silane/siloxane emulsion remained almost fully intact. The strain increased very slowly, and the average value did not exceed 0.34 mm/m, which was the best performance among all groups. Therefore, specimens admixed with silane/siloxane emulsions had the most ASR-stable properties, even if undergoing the very harsh cyclic exposures to NaCl solutions with concentrations up to 10%.
The results for the rhyolite aggregates in Figure 7,
right, were also encouraging. The control specimens again showed a steady increase of average strain which was significantly milder than that of the graywacke control group due to the lower reactiveness of rhyolite with alkali. The average strain value after 10 exposure cycles was 0.42 mm/m, without exceeding the 0.5 mm/m limit. The specimens admixed with a plain silane produced lower expansion levels than the control specimen. After 10 exposure cycles, the average strain value reached 0.29 mm/m. This shows that silanes can effectively prevent ASR-induced expansions in cementitious systems containing rhyolite aggregates. Best among all, however, was the silane/siloxane emulsion specimen. It hardly produced any volume increase during the entire test period. The average strain value after 10 cycles reached 0.04 mm/m, which is more than ten times below the official expansion limit and far below the control specimen values. This is very convincing evidence that the silane/siloxane emulsions can effectively help to inhibit ASR in specimens containing rhyolite aggregates even undergoing cyclic exposures to 10% NaCl solutions.
3.3 Further discussion
The above results clearly demonstrate that organosilicon chemicals can effectively inhibit the ASR-induced expansion in concrete, even if the concrete contains highly alkali-sensitive aggregates and/or excessive alkali supplies are present. The reason for this beneficial effect results from the hydrophobic properties of siloxane- and silane-based agents which come into play. ASR issues occur as soon as alkali and reactive silica are present and form gel products inside the concrete. However, if there is no or only limited access to moisture, the ASR gel will not be subject to swelling which is the root cause for detrimental stress build-up and cracking inside the concrete mass.
As shown above, organosilicon admixtures such as silanes or siloxanes can render the cementitious matrix highly hydrophobic. Consequently, any potential pathways to ASR gels inside the matrix can be hydrophobized, preventing moisture from getting into contact with the swellable gels. Even pathways formed at later stage, e.g. microcracks related to ongoing thermal or mechanical stress, remain water repellent, owing to the integral nature of the hydrophobic treatment. Hence, ASR-induced damage can be avoided reliably.
By this means, the use of organosilicon admixtures is a preventive measure, which is especially advantageous when ASR-unstable raw materials are encountered in real life applications. More-over, organosilicon admixtures create sufficiently stable bonds with cementitious phases, even under harsh chemical or physical conditions. This further enhances the long-term protection against ASR effects, as the hydrophobic performance can be well preserved over an extended period of time. A straightforward example is presented in Figure 8.
In the untreated specimen (Figure 8, left), major through cracks have developed in the grains of the coarse aggregate, due to the ASR effect. While in the treated specimen (Figure 8, right), hardly any cracks have formed in the matrix.
4 Summary
Organosilicon hydrophobic agents are highly versatile and yet unique chemicals which enable extraordinary waterproofing performance in cementitious materials. Due to their exceptional chemical and physical stability, they can achieve particularly effective water repellency and can significantly enhance concrete durability even under very harsh conditions. In addition to surface treatment applications, organosilicon agents can also be used as integral waterproofing agents. In this case, their protection against undesired water penetration into the cementitious mass is more distinct and more durable. Furthermore, the integral use of organosilicon agents can provide further beneficial effects against water-associated deterioration processes such as the Alkali Silica Reaction. Such products are able to enhance the ASR stability of concrete, even if ASR-unstable raw materials are present in the cementitious matrix.
//www.wacker.com" target="_blank" >www.wacker.com:www.wacker.com
p. 14
Association, 1997
Vermeidung von Schäden an Fahrbahndecken aus Beton in Folge von Alkali-Kieselsäure-Reaktion (AKR), Bonn, 2013